MedTech News
.................... by Andrew Celentano

Prostate cancer cells use unique metabolic pathway to thrive in bone tissue
A University of Kentucky Markey Cancer Center study reveals how prostate cancer cells adapt their metabolism to thrive in bone tissue, offering a potential new treatment target for patients with advanced disease.
Cancer’s hidden ‘safety switch’: Silencing TAK1 gene could boost immunotherapy performance
Australian researchers have discovered that the TAK1 gene helps cancer cells survive attack from the immune system, revealing a mechanism that may limit the effectiveness of immunotherapy treatments.
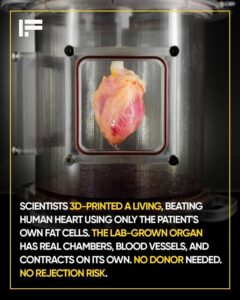
Scientists 3D print a living, beating human heart
Researchers at Tel Aviv University achieved a medical milestone once thought impossible: creating a living, beating human heart using a patient’s own fat cells.

Zoll launches 5th-gen wearable cardioverter defibrillator
Zoll announced today that it launched its next-generation LifeVest wearable cardioverter defibrillator (WCD) in the U.S.
New Raman imaging system detects subtle tumor signals
Researchers have developed a new compact Raman imaging system that is sensitive enough to differentiate between tumor and normal tissue. The system offers a promising route to earlier cancer detection and to making molecular imaging more practical outside the lab.

Hidden ‘switches’ in DNA reveal new insights into Alzheimer’s disease
A tiny percentage of our DNA—around 2%—contains 20,000-odd genes. The remaining 98%—long known as the non-coding genome, or so-called ‘junk’ DNA—includes many of the “switches” that control when and how strongly genes are expressed.
Cutera® Announces New FDA Clearances for truFlex® to Support Rehabilitation, Recovery and Muscle Wellness
The next evolution of truFlex fortifies its position as a versatile solution for rehabilitation with new applications in functional strength for the abdomen, arms, calves, glutes and thighs.
RIVANNA announces FDA clearance of Accuro 3S Needle Guide Kit
The Accuro 3S Needle Guide Kit, designed for use with the Accuro 3S ultrasound imaging system and Dual-Array transducer, includes single-use, sterile components that support infection control and promote workflow efficiency
