MedTech News
.................... by Andrew Celentano
FDA clears Techsomed BioTraceIO360 for kidney ablation
Techsomed this week announced it received FDA 510(k) clearance to expand indications for its BioTraceIO360 software platform to include percutaneous ablation of soft tissue in the kidney.
Brainlab unveils FDA-approved spine mixed reality navigation in US
The system leverages the company’s optical navigation system technology and the latest advancements in mixed reality.

Medtronic gets FDA nod for implantable neuromod for urinary incontinence
Medtronic (NYSE: MDT)+
announced today that it received FDA approval for its Altaviva neuromodulation device.

Breakthrough for Organ Transplants May Be Realized by Turning Organs into Glass
Discover how turning organs into glass could revolutionize crucial transplant logistics.
IceCure Granted Notice of Allowance for U.S. Patent for its Next-Generation XSense™ Cryoprobes
CAESAREA, Israel, Sept. 18, 2025 /PRNewswire/ — IceCure Medical Ltd. (NASDAQ: ICCM) (“IceCure”, “IceCure Medical” or the “Company”), developer of minimally-invasive cryoablation technology that destroys tumors by freezing as an option to surgical tumor removal, today announced it has received a Notice of Allowance from the U.S. Patent and Trademark Office for its patent titled “Cryoprobe”.

J&J launches IVL device in Europe; Medtronic looks at pacing for more patients
J&J’s Shockwave Javelin intravascular lithotripsy catheter treats people with peripheral artery disease.
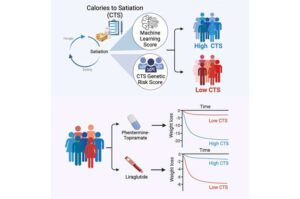
Genetic test predicts response to weight-loss drugs
Mayo Clinic researchers have developed a genetic test that can help predict how people will respond to weight loss medications such as GLP-1s.
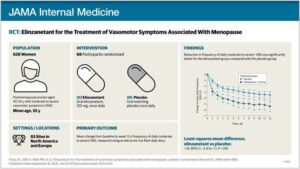
Menopause drug reduces hot flashes by more than 70%, international clinical trial finds
The investigational drug elinzanetant significantly reduces hot flashes and night sweats for postmenopausal women, a large, international clinical trial has found.
