MedTech News
.................... by Andrew Celentano
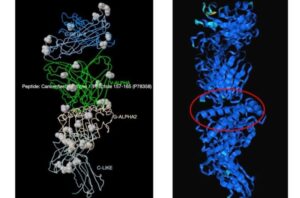
AI approach paves way for smarter T-cell immunotherapy and vaccine development
Researchers have harnessed the power of artificial intelligence to tackle one of the most complex challenges in immunology: predicting how T cells recognize and respond to specific peptide antigens.
Blood test could streamline early Alzheimer’s detection in underrepresented populations
In a landmark study of Hispanic and Latino adults, researchers at University of California San Diego School of Medicine have identified a link between self-reported cognitive decline and blood-based biomarkers, which could pave the way for a simple blood test to help diagnose Alzheimer’s disease and related dementias.
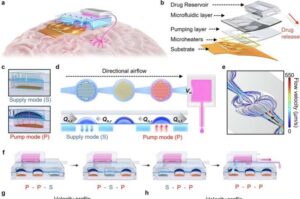
Wireless neural interface could be used for drug delivery to deep brain regions
A research team has developed an implantable wireless neural interface capable of delivering drugs precisely to deep regions of the brain.

Soundscapes from text: VR system personalizes PTSD exposure therapy with rapid audio generation
Post-traumatic stress disorder (PTSD) is a mental health disorder triggered by traumatic experiences such as accidents, disasters, or violence. Exposure therapy, a scientifically validated treatment for PTSD, involves the gradual re-exposure of patients to trauma-related cues to reduce anxiety and avoidance behaviors.
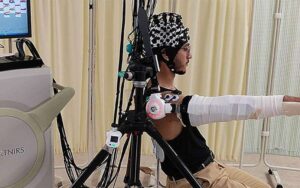
Cyborg-type robots can boost neuroplasticity when users control their own movement
Researchers at University of Tsukuba have demonstrated for the first time that brain regions responsible for high-level motor planning and preparation, such as the premotor cortex, are activated when a cyborg-type robot is volitionally controlled.

NHS offers artificial pancreas to pregnant women with type 1 diabetes in the UK
The NHS announced a first-of-its-kind initiative in England, offering automated insulin delivery systems for pregnant women with diabetes.

Pulse Biosciences wins FDA IDE to conduct study of nsPFA tech for AFib
Pulse Biosciences (Nasdaq:PLSE) announced today that the FDA granted investigational device exemption (IDE) for its nanosecond pulsed field ablation (nsPFA) system.

Lifeward wins CE mark for ReWalk 7 personal endoskeleton
Lifeward (Nasdaq:LFWD) announced today that it received CE mark approval for its ReWalk 7 personal exoskeleton.
