MedTech News
.................... by Andrew Celentano

SoClean wins FDA de novo nod for CPAP, ventilation hose cleaner
SoClean this week announced it was granted FDA de novo clearance for its at-home CPAP mask and ventilation hose cleaner SoClean 3+.

Si-Bone wins FDA clearance, breakthrough nod for pelvic fixation implant
Si-Bone this week announced it received FDA 510(k) clearance and FDA breakthrough device designation for its iFuse Torq TNT Implant System.

FDA approves label expansion for Abbott HeartMate 3 LVAD to remove aspirin from treatment regimen
Abbott (NYSE: ABT)+
announced today that the FDA approved a label change for its HeartMate 3 left ventricular assist device (LVAD).
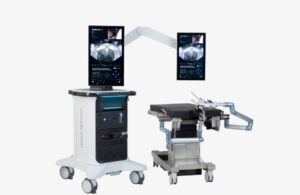
FDA clears next-gen surgical robot from Procept Biorobotics
Procept BioRobotics (Nasdaq:PRCT) announced today that it received FDA 510(k) clearance for its next-generation Hydros robotic surgery system.

AlgoDx receives FDA clearance for sepsis detection software
AlgoDx, a leading medical AI company with EU and UK-approved ML-based software for sepsis prediction in commercial use, has announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its sepsis detection software using a proprietary algorithm, NAVOY CDS. This milestone marks a major step forward in AlgoDx’s expansion into the U.S. market.

New ultrasonic device launches to aid with vision loss after stroke and brain injury
Ocutech, Inc., in collaboration with electrical, software and mechanical engineering experts, has announced the limited first release of a new ultrasonic sensor device

Enovis unveils new ankle replacement system
Enovis (NYSE: ENOV)+ announced today that it unveiled its STAR Ankle system with new e+ Polyethylene.
Delcath Gets FDA Approval for Cancer Drug-Device Combo 10 Years After Rejection
Following a nearly decade-long effort, Delcath Systems finally won the FDA’s greenlight for its Hepzato Kit for the liver-directed treatment of adult patients with metastatic uveal melanoma.
