MedTech News
.................... by Andrew Celentano

Electric bandage holds promise for treating chronic wounds
Researchers have developed an inexpensive bandage that uses an electric field to promote healing in chronic wounds

Researchers develop a new vaccine additive that creates a stronger, tunable immune response
Researchers at Stanford Engineering have developed a nanoparticle platform that could make existing vaccines more effective, including those for influenza, COVID-19, and HIV.
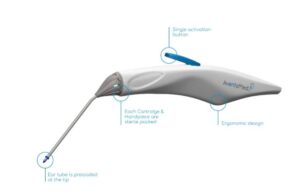
Karl Storz’s AventaMed wins FDA nod for ear tube placement device
AventaMed, a Karl Storz company, announced that it received FDA 510(k) clearance for its Solo+ ear tube placement system.

Medtronic wins FDA nod for next-gen Simplera CGM
Medtronic (NYSE: MDT)+ got another boost for its CGM portfolio, adding to the collaboration with Abbott announced today.
Johnson & Johnson MedTech Announces Labeling Amendment of LINX™ Reflux Management System in the U.S.
CINCINNATI, OH August 7, 2024 – Ethicon*, a Johnson & Johnson MedTech company**, announced today that the U.S. Food & Drug Administration (FDA) has approved a label update to expand the availability of the LINX™ Reflux Management System to include patients with Barrett’s esophagus (BE) experiencing gastroesophageal reflux disease (GERD) symptoms.

Aspen Medical Products Launches First Bracing Solution Available for PJK Mitigation
IRVINE, Calif., Aug. 6, 2024 /PRNewswire/ — Aspen Medical Products (Aspen), the industry leader in spine solutions for pain and mobility management, has launched the Align Orthosis. In collaboration with spine surgeon Han Jo Kim, MD of the Hospital for Special Surgery, the all-new Align Orthosis is the first prefabricated TLSO designed to help mitigate post-surgical incidence of proximal junctional kyphosis (PJK).
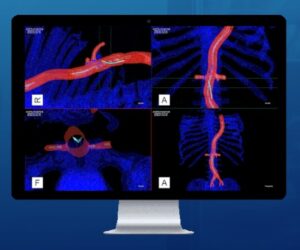
Centerline Biomedical receives FDA 510(k) clearance for new IOPS® Viewpoint Catheter, further addressing market need to reduce physician exposure to harmful x-ray radiation during image-guided, endovascular procedures
CLEVELAND, Aug. 6, 2024 /PRNewswire/ — Centerline Biomedical, Inc. (“Centerline”), an innovation leader in cardiovascular navigation and visualization systems, announced today that the IOPS Viewpoint Catheter has received US Food and Drug Administration (FDA) 510(k) clearance. The Viewpoint Catheter is the most recent addition to the company’s patented IOPS (Intra-Operative Positioning System) portfolio.
New alloy shown to produce better human joint implants
Stiffness, pain and infections in orthopedic surgery is being tackled by Flinders University researchers driving innovation in alloy materials to produce safe and superior implants compatible with human tissue.
