MedTech News
.................... by Andrew Celentano

New ‘molecular flashlight’ technique detects brain metastases in mice using ultra-thin light probe
Monitoring molecular changes in the brain caused by cancer and other neurological pathologies in a non-invasive way is one of the major challenges in biomedical research. A new experimental technique has achieved this by introducing light into the brains of mice using an ultra-thin probe.
Autonomix secures U.S. patent for cancer-treating tech
Autonomix today announced it received a patent from the U.S. Patent and Trademark Office (USPTO) for its catheter-based cancer tumor treating technology.

New laser technology improves stent effectiveness in vascular treatments
A Korea Institute of Science and Technology (KIST) research team has developed a novel stent surface treatment technology using laser patterning. This technology promotes endothelial cell growth while inhibiting smooth muscle cell dedifferentiation in blood vessels.
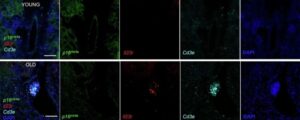
Novel biomarker catches aging cells in the act
Mayo Clinic researchers have identified interleukin-23 receptor (IL-23R) as a significant biomarker of cellular senescence and aging in both mice and humans. Experiments show that IL-23R levels in the bloodstream increase with age and can decrease, reflecting senescent cell clearing, with senolytic therapies.
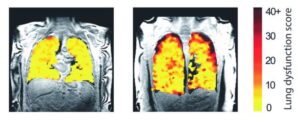
New lung scanning method can show treatment effects in real time
A new method of scanning lungs is able to show the effects of treatment on lung function in real time and enable experts to see the functioning of transplanted lungs. This could enable medics to identify any decline in lung function sooner.
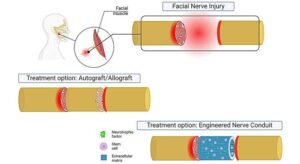
Engineering a smile: Stem cell–based conduits restore facial nerve function in animal study
A gesture as simple as a smile can often convey what words cannot. This is part of why nonverbal communication is so central to human interaction. It is also why facial nerve disorders and injuries can be devastating.
Engineered T cells could help patients overcome resistance to CAR T cell therapy
Physician-researchers with City of Hope have developed a way to add features to T cells to help them overcome mechanisms of chimeric antigen receptor (CAR) T cell therapy resistance. Their new system is outlined in a paper published in Nature Biomedical Engineering.
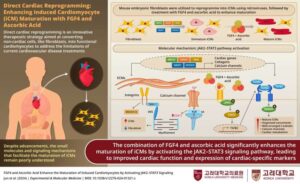
Reprogramming cells for heart repair: New method transforms ordinary fibroblasts into mature cardiomyocytes
Cardiovascular disease continues to lead as the primary cause of death across the globe, taking millions of lives every year. Damage caused by these diseases is particularly difficult to repair, since the heart has minimal ability to regenerate itself. But what if we could reprogram the body’s own cells to restore damaged tissue? This question has been tackled by scientists at Korea University, led by Dr. Myeong-Hwa Song.
