MedTech News
.................... by Andrew Celentano

FDA clears Orthofix Fitbone transport and lengthening system
Orthofix (Nasdaq: OFIX) today announced it received FDA 510(k) clearance for the Fitbone Transport and Lengthening System and reported the first implant of the device.

Cordis wins FDA nod for vascular closure device
Cordis announced today that the FDA granted premarket approval (PMA) for its Mynx Control venous vascular closure device (VCD).
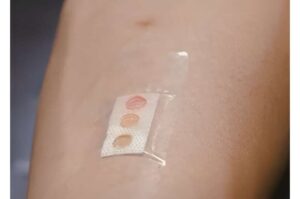
Scientists develop bandage that measures glucose levels in sweat using microlaser technology
Scientists from Nanyang Technological University, Singapore (NTU Singapore) have developed a band-aid or plaster that measures body biomarkers that can indicate health or disease through sweat, paving the way for a new non-invasive and effective way for patients to monitor their health.

Pulse Biosciences wins FDA breakthrough nod for AFib-treating pulsed field ablation tech
Pulse Biosciences (Nasdaq:PLSE) announced today that the FDA granted breakthrough device designation for its pulsed field ablation system. Shares of PLSE rose more than 20% in mid-morning trading on the back of this announcement.

New wearable patch measures trace perspiration during activity and rest
Researchers at University of Tsukuba have pioneered a state-of-the-art wearable device capable of precisely and continuously measuring trace perspiration amounts. This innovative device is adept at monitoring perspiration levels during not only physical activity but also rest. Its potential applications extend beyond monitoring dehydration caused by exercise or heat exposure, encompassing broader domains such as daily health management and disease detection.
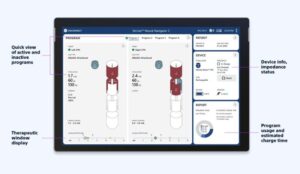
Boston Scientific obtains CE mark for image guided programming software for deep brain stimulation
The Vercise Neural Navigator 5 with STIMVIEW XT technology is designed to streamline procedures for people living with neurological conditions according to the company.
Signum Surgical wins FDA de novo nod for bioabsorbable implant
Signum Surgical announced today that the FDA granted de novo clearance for its BioHealx implant for treating anal fistula.

Implantable microphone could lead to fully internal cochlear implants
This tiny, biocompatible sensor may overcome one of the biggest hurdles that prevent the devices from being completely implanted.
