MedTech News
.................... by Andrew Celentano
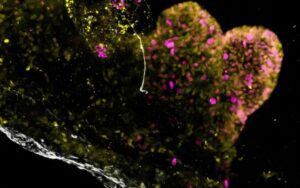
Lab-grown mini-stomachs could boost understanding of rare diseases
Researchers at UCL and Great Ormond Street Hospital (GOSH) have developed the first-ever lab-grown mini-stomach that contains the key components of the full-sized human organ.

First-of-its-kind probe monitors fetal health in utero during surgery
Northwestern University researchers have developed the first device that can continuously track a fetus’s vital signs while still in the uterus.

Pulsenmore and Clalit Health Services Launch First At-Home Follicular Monitoring Service for IVF and Fertility Preservation
RAMAT GAN, Israel, Jan. 26, 2026 /PRNewswire/ — Pulsenmore Ltd. (NASDAQ: PLSM) (TASE: PLSM), a global leader in connected home ultrasound technology, today announced an additional strategic commercial agreement with Clalit Health Services, Israel’s largest health maintenance organization (HMO), to supply Pulsenmore FC, a new at-home ultrasound solution designed for remote follicular monitoring in women undergoing fertility care (IVF and fertility preservation).
Amend Surgical Proudly Announces ISO 13485 Certification, Advancing Toward Launch of Amend Tissue Tape™
ALACHUA, Fla., Jan. 26, 2026 /PRNewswire/ — Amend Surgical, a medical device company focused on novel products for oral wound-care and regenerative biomaterials, today announced that it has achieved ISO 13485:2016 certification, the global standard for medical device quality management systems.

Spine Innovation Announces FDA 510(k) Clearance for LOGIC™ Titanium Expandable Interbody System
CORONADO, Calif., Jan. 26, 2026 /PRNewswire/ — Spine Innovation, LLC, a medical device startup that develops novel interbody fusion implants, announced today that is has received FDA 510(k) clearance to market the LOGIC™ Titanium Implant System.

MannKind earns expanded FDA labeling for inhaled insulin
MannKind (Nasdaq:MNKD) announced today that it received FDA approval for an update to the prescribing information for Afrezza inhaled insulin.

HighLife wins CE mark for TMVR system
HighLife announced today that it received CE mark approval for its transcatheter mitral valve replacement (TMVR) system.

Penumbra launches next-gen Lightning Flash 3.0 thrombectomy system
Penumbra (NYSE:PEN) announced today that it launched the Lightning Flash 3.0 computer-assisted vacuum thrombectomy (CAVT) system.
