
Pulse Biosciences touts first-in-human pulsed field ablation cases
Pulse Biosciences (Nasdaq:PLSE) today announced the first completed cases in a first-in-human feasibility study of its pulsed field ablation system.

Pulse Biosciences (Nasdaq:PLSE) today announced the first completed cases in a first-in-human feasibility study of its pulsed field ablation system.
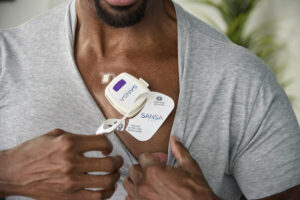
ATLANTA, Aug. 7, 2024 /PRNewswire/ — Huxley Medical, a developer of technologies that streamline cardiopulmonary care, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its groundbreaking, chest-worn, sleep apnea diagnostic patch, SANSA.
CINCINNATI, OH August 7, 2024 – Ethicon*, a Johnson & Johnson MedTech company**, announced today that the U.S. Food & Drug Administration (FDA) has approved a label update to expand the availability of the LINX™ Reflux Management System to include patients with Barrett’s esophagus (BE) experiencing gastroesophageal reflux disease (GERD) symptoms.

Stryker (NYSE: SYK)+
announced today that it launched its Pangea plating system portfolio of devices.
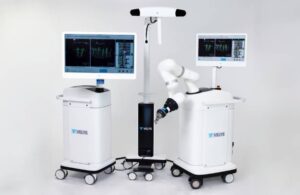
Johnson & Johnson’s (NYSE: JNJ)+
DePuy Synthes announced today that it launched a dual-use robotics and standalone navigation platform that uses its Velys robot in spine surgery.

A paraplegic person carried the Olympic torch as part of the traditional relay using an exoskeleton developed by Wandercraft.

RA’ANANA, Israel, July 11, 2024 /PRNewswire/ — Inspira™ Technologies OXY B.H.N. Ltd. (Nasdaq: IINN) (Nasdaq: IINNW) (the “Company” or “Inspira”), a breakthrough medical technology company, is excited to announce the receipt of the Israeli Ministry of Health’s medical devices and accessories (“AMAR”) approval for the INSPIRA™ ART100, an Extra-Corporeal Membrane Oxygenation and Cardiopulmonary Bypass system. This is a pivotal milestone in Inspira’s strategy to conduct business development activities to bring its innovative products and technologies to the market.

Front Line Medical Technologies has announced that its COBRA-OS (Control Of Bleeding, Resuscitation, Arterial Occlusion System) has officially been granted CE marking under the new European Medical Device Regulations.
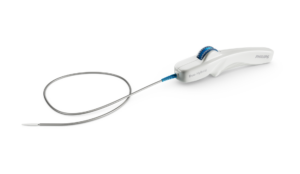
Philips, a global leader in health technology, has announced the first implant of the Duo Venous Stent System, an implantable medical device indicated to treat symptomatic venous outflow obstruction in patients with chronic venous insufficiency (CVI), following premarket approval (PMA) from the U.S. Food and Drug Administration (FDA).

This marks a significant advancement in the international progress of VenusP-Valve and represents another milestone following its CE MDR approval in April 2022.