
VUNO’s AI Cardiac Risk System Gains CE MDR and UKCA Certifications
Milestone Approvals Enable European and UK Market Access for AI-Based Cardiac Arrest Prediction Tool

Milestone Approvals Enable European and UK Market Access for AI-Based Cardiac Arrest Prediction Tool

New Liquid Biopsy Technology Offers Hope in Diagnosing One of the Deadliest Cancers at an Early Stage

Two meters of DNA is crammed into the nucleus of every human cell. Bin Zhang wants to know how gene expression works in this minuscule space.
This historic acquisition reduces barriers that prevent people from taking control of their lifelong health. Full Body MRI scan now takes just 22 minutes instead of 60 minutes, costs $499 instead of over $1,500
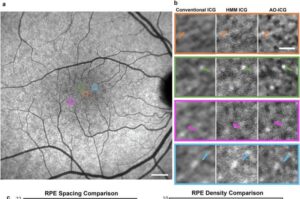
Scientists at the National Institutes of Health (NIH) have leveraged artificial intelligence to transform a device designed to see tissues in the back of the eye into one sharp enough to make out individual cells. The technique provides imaging resolution that rivals the most advanced devices available and is cheaper, faster, and doesn’t require specialized equipment or expertise. The strategy has implications for early detection of disease and for the monitoring of treatment response by making what was once invisible now visible.
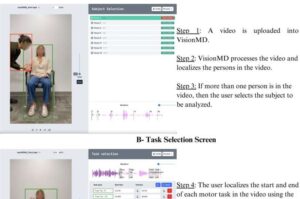
A University of Florida researcher has developed an open-source computer program that uses artificial intelligence to analyze videos of patients with Parkinson’s disease and other movement disorders. The tool, called VisionMD, helps doctors more accurately monitor subtle motor changes, improving patient care and advancing clinical research.

U-Pump announced on social media that it reached a new milestone in its development of an AI-powered, reusable insulin patch pump.

Researchers designed MAIJU (Motor Assessment of Infants with a Jumpsuit) to solve these problems. The suit is based on multisensor measurements, which are used to make reliable and versatile measurements of children’s motion with the help of dedicated AI algorithms.

University of Virginia School of Medicine scientists have created a computational tool to accelerate the development of new disease treatments. The tool goes beyond current artificial intelligence (AI) approaches by identifying not just which patient populations may benefit but also how the drugs work inside cells.

Dr. George Shaker has huge hopes for a small radar device he developed with a team of researchers at the University of Waterloo. An engineer who has pursued the possibilities of radio waves for more than 20 years, Shaker envisions the installation of the silent, unobtrusive system almost everywhere people sit to help detect heart problems before they become life-threatening.