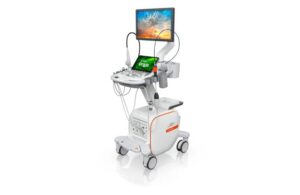
FDA clears Siemens Healthineers’ AI-powered cardiovascular ultrasound and catheter
Siemens Healthineers announced today that it received FDA clearance for its Acuson Origin ultrasound system and AcuNav Lumos 4D ICE catheter.

Siemens Healthineers announced today that it received FDA clearance for its Acuson Origin ultrasound system and AcuNav Lumos 4D ICE catheter.
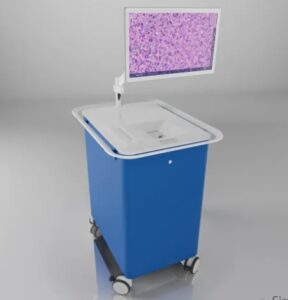
SANTA CLARA, Calif., Aug. 9, 2024 /PRNewswire/ — Invenio Imaging, a leader in intraoperative fresh tissue imaging and artificial intelligence (AI), announced today the enrollment of the first patients in a US pivotal study of its AI-based image analysis module for lung cancer

MIDDLETON, Wis., Aug. 7, 2024 /PRNewswire/ — Natus Medical Incorporated has announced the launch of autoSCORE, the first-of-its-kind artificial intelligence model capable of automatic and comprehensive clinical EEG interpretation, providing accuracy on par with medical experts.

Hyperfine (Nasdaq:HYPR) announced today that the FDA cleared its ninth-generation AI-powered Swoop system software.
Bern & Boston, US, June 24th, 2024 – Ikerian AG (“Ikerian”) a leader in developing software solutions for medical image and data management and artificial intelligence (AI) in healthcare, today announced the registration as Class IIa medical devices under the European Union Medical Device Regulation (Regulation (EU) 2017/745) (EU-MDR) of 4 devices.

Powered by this AI, the Pocket-Sized Kardia 12L ECG System Enables Faster, Easier Detection of Life-Threatening Cardiac Conditions

Leuko, founded by a research team at MIT, is giving doctors a noninvasive way to monitor cancer patients’ health during chemotherapy — no blood tests needed.
Medical imaging AI company receives approval for algorithm enabling opportunistic detection and triage of vertebral compression fractures.
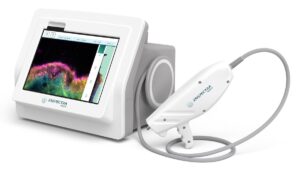
Enspectra Health, a health tech company, today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for its next generation AI-powered VIO™ Skin Platform (VIO)

Profound Medical (Nasdaq:PROF) announced today that it received FDA 510(k) clearance for its second AI model for treating prostate cancer.