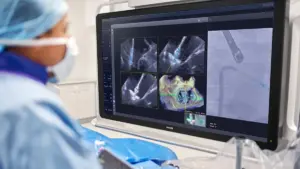
Philips wins FDA clearance for imaging device developed with Edwards
The system uses AI to optimize the placement of Edwards’ mitral valve repair device.

The system uses AI to optimize the placement of Edwards’ mitral valve repair device.

An Abbott (NYSE:ABT) official has shared a significant reimbursement win for its Esprit BTK drug-eluting scaffold system.

Noah Labs Vox uses AI algorithms to detect worsening heart failure based on patients’ voice recordings.

Achieving MDR compliance shows Modius Sleep meets safety, clinical, and quality standards required for entry into the EU market.

BEIJING, March 26, 2026 /PRNewswire/ — Wingderm®, a manufacturer of medical aesthetic devices, announces that its Lasermach system has received expanded clearance from the U.S. Food and Drug Administration (FDA). With this latest clearance, Lasermach now achieves FDA clearance across all three wavelengths.

Inspire Medical (NYSE:INSP) announced today that it received expanded FDA approval for its Inspire IV and V neuromodulation systems.
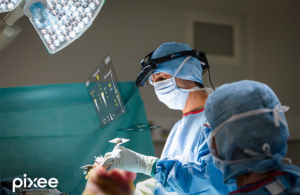
Pixee Medical announced today that it received CE mark approval for Knee+ NexSight, with the first cases already performed in Europe.
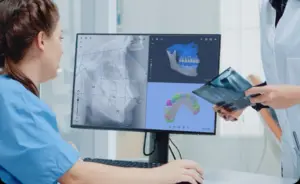
DOVER, Del., March 24, 2026 /PRNewswire/ — Orca Dental AI, the global leader in AI-powered orthodontic imaging solutions and creator of the CephX platform, today announced it has received FDA clearance for 3D imaging, marking a major milestone in the company’s mission to bring advanced AI-powered diagnostic support into everyday clinical workflows.

MOUNTAIN LAKES, N.J., March 24, 2026 /PRNewswire/ — LiviWell, a women’s health company dedicated to developing innovative solutions for intimate health, today announced that the U.S. Food and Drug Administration (FDA) has cleared Livi, a first-of-its-kind device designed to support vaginal health by absorbing semen and other post-intercourse fluids.

GE HealthCare (Nasdaq:GEHC) announced today that it received FDA 510(k) clearance for its Photonova Spectra solution.