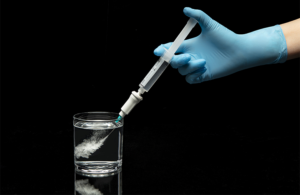
Agitated Solutions wins CE mark for cardiac microbubble generator device
Agitated Solutions announced that it received CE mark approval for its novel Orbis microbubble generator device.

Agitated Solutions announced that it received CE mark approval for its novel Orbis microbubble generator device.

Zeta Surgical announced today that the FDA granted 510(k) clearance for its Zeta Navigation System and its navigated Stylet and Bolt instruments.
The U.S. Food and Drug Administration has approved Merck’s Idvynso (doravirine/islatravir), a new, once-daily, two-drug single tablet for the treatment of HIV-1 infection in adults to replace the current antiretroviral regimen in those who are virologically suppressed (HIV-1 RNA <50 copies per mL).

Establishing it as the first ever reusable intermittent urinary catheter system and creating a new device classification in an industry that exclusively relied on single-use catheters for decades

Alzheimer’s disease agitation could mean peak sales of over $2.1 billion for Axsome’s Auvelity, according to analysts at William Blair.
The TAVVE study will begin later this year with ten patients, whose 30-day safety results will be submitted to the FDA.
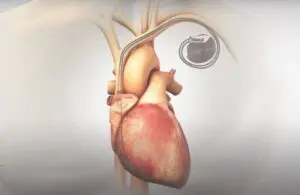
Orchestra BioMed (Nasdaq:OBIO) announced today that it received a second FDA breakthrough device designation for its AVIM therapy.

GRAND RAPIDS, Mich., April 30, 2026 /PRNewswire/ — Capitan Orthopedics, Inc. (https://capitanortho.com/) announced today that it has been granted Breakthrough Device Designation from the U.S. Food & Drug Administration (FDA) for its SupraSpacerTM implant.

FDA authorization marks critical milestone enabling advancement of next-generation bone regeneration therapy for patients with degenerative spinal diseases
Avatar’s software translates CT and MR imaging data into 3D images to streamline surgical planning and operating room workflows.