Medtronic wins FDA nod for next-gen mitral valve
Medtronic (NYSE:MDT) announced today that it received FDA approval for its Mosaic Neo mitral bioprosthesis and began its U.S. launch.

Medtronic (NYSE:MDT) announced today that it received FDA approval for its Mosaic Neo mitral bioprosthesis and began its U.S. launch.

Earflo announced that it received FDA clearance for its non-invasive device designed to treat negative middle ear pressure in children.
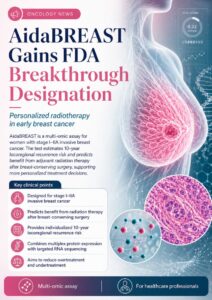
LAGUNA HILLS, Calif., April 29, 2026 /PRNewswire/ — Prelude Corporation (PreludeDx™), a leader in precision diagnostics for early-stage breast cancer, announced today that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device designation for the AidaBREAST® test.

HERSHEY, Pa., April 29, 2026 /PRNewswire/ — Chest Wall Innovations, a medical device company focused on advancing the standard of care in chest wall surgery, today announced that the U.S. Food and Drug Administration (FDA) granted 510(k) clearance for its PC Fix Rib Fixation System on April 24, 2026, clearing the path for U.S. commercialization.
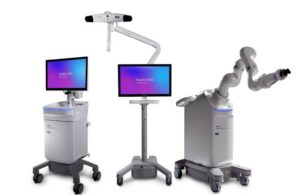
Medtronic (NYSE:MDT) announced today that it received CE mark approval for its Stealth AXiS surgical robot platform.
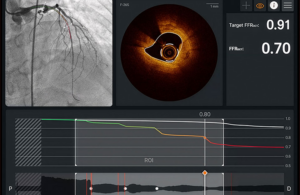
Abbott (NYSE:ABT) announced today that it received FDA clearance and CE mark for its next-generation Ultreon 3.0 software.
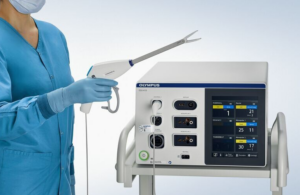
Olympus announced today that it received FDA 510(k) clearance for its Powerseal open extended jaw sealer/divider for use in open surgery
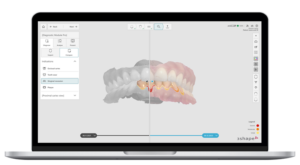
COPENHAGEN, Denmark, April 28, 2026 /PRNewswire/ — 3Shape today announced that the U.S. Food and Drug Administration (FDA) has granted FDA clearance for its Dx Software following a review completed on April 10. 2026.
IRVINE, Calif., April 28, 2026 /PRNewswire/ — Laguna Diagnostics, LLC today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation to the company’s mRNA Gene Biomarker Test, a novel blood-based test designed to aid in the differentiation of schizophrenia and bipolar I disorder in symptomatic patients.
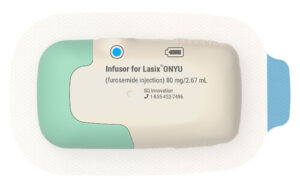
BURLINGTON, Mass., April 28, 2026 /PRNewswire/ — SQ Innovation, developer of Lasix® ONYU, today announced two important regulatory milestones in its international expansion strategy: the receipt of a CE marking certificate for the Lasix ONYU device, and the acceptance of Lasix ONYU by the European Medicines Agency (EMA) for review under the centralized procedure on the basis of technical innovation. Together, these milestones represent a significant step forward in bringing this novel at-home subcutaneous furosemide treatment to patients in Europe and other international markets.