
Motif Neurotech wins FDA IDE to evaluate brain implant for treating depression
Motif Neurotech announced today that it received FDA investigational device exemption (IDE) to evaluate its brain stimulation implant.

Motif Neurotech announced today that it received FDA investigational device exemption (IDE) to evaluate its brain stimulation implant.
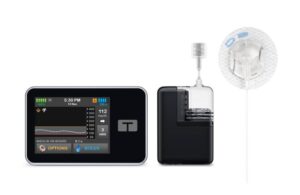
Tandem Diabetes Care (Nasdaq:TNDM) today announced FDA clearance for its automated insulin delivery (AID) technology for use in pregnancy.

NEW YORK, April 27, 2026 /PRNewswire/ — TytoCare, a virtual care company enabling accessible, high-quality primary care from home, today announced it has received FDA De Novo classification for the first model of Tyto Insights™ for ENT Suite, marking the creation of a new regulatory category for AI-powered ENT image analysis.
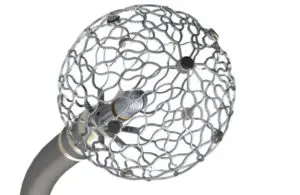
Medtronic (NYSE:MDT) today announced new milestones for its Affera family of technologies, including FDA breakthrough device designation.

BrioHealth announced that it received conditional FDA approval to conduct a clinical evaluation of its ventricular assist device (VAD) in pediatric patients.
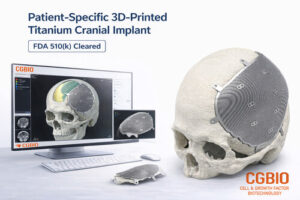
SEOUL, South Korea, April 24, 2026 /PRNewswire/ — CGBIO (CEO: Hyunseung Yu) announced that its patient-specific titanium implant, EASYMADE TI, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) on April 9. The FDA 510(k) pathway is a premarket clearance process that demonstrates substantial equivalence to a legally marketed predicate device, allowing commercialization in the United States.

Medtronic (NYSE:MDT) announced today that it received CE mark approval for its ApexCut surgical blades for ENT procedures.

Approved Thursday via the FDA’s Commissioner’s National Priority Voucher program, Otarmeni is the first gene therapy for hearing loss—and the first treatment to target an underlying cause of the condition.

Biotronik announced today that it received FDA approval for its Acticor Sky and Rivacor implantable defibrillator systems.

The newly approved HIV drug Idvynso will also help Merck diversify as loss of exclusivity looms over its top-selling product, the mega-blockbuster cancer drug Keytruda.