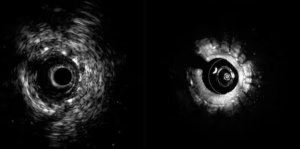
Conavi wins FDA clearance for intravascular ultrasound platform
Conavi Medical announced today that it received FDA 510(k) clearance for its next-generation hybrid imaging system.

Conavi Medical announced today that it received FDA 510(k) clearance for its next-generation hybrid imaging system.

Fentanyl Urine Visual Test is available through Labcorp Point of Care, previously known as MEDTOX Diagnostics

MONTREAL, April 17, 2026 /PRNewswire/ — Bracco Imaging, a global leader in diagnostic imaging, today announced that Health Canada approved VUEWAY® injection (gadopiclenol), a macrocyclic gadolinium-based contrast agent (GBCA), for use in contrast-enhanced magnetic resonance imaging (CE-MRI).
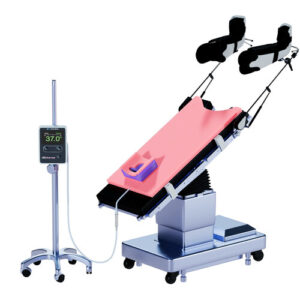
PITTSBURGH, April 17, 2026 /PRNewswire/ — Xodus Medical today announced the launch of the Hot Pink Pad®, a next-generation patient temperature management and positioning system designed to improve surgical outcomes, enhance patient safety, and streamline operating room workflows.
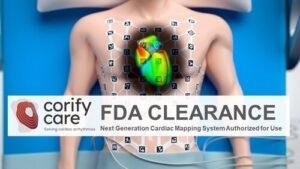
MADRID, April 17, 2026 /PRNewswire/ — Corify Care today announced that the U.S. Food and Drug Administration (FDA) has granted clearance for the ACORYS® System, the first imageless, non-invasive cardiac mapping platform capable of delivering a global, four-chamber view of cardiac electrical activity in real-time.

Aneuvo announced that it received FDA clearance for its ExaStim spinal cord stimulation (SCS) system

The CYBORG study aims to assess the efficacy, safety and usability of the system.
AI will soon enable clinicians to predict ovulation and optimize IVF cycle timing, improving outcomes and operational efficiency.
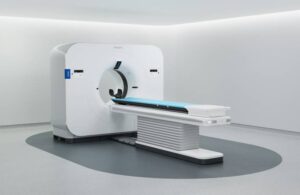
Philips (NYSE:PHG) announced today that it received FDA 510(k) clearance for its AI-powered Spectral CT Verida system.
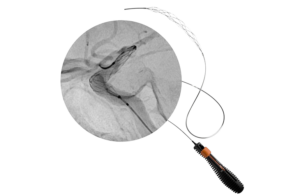
Rapid Medical announced today that it received FDA clearance for the Tigertriever 25 thrombectomy device, its latest addition to the platform.