
OncoPatch wins FDA nod for tumor-treating patch device
OncoPatch announced today that it received FDA 510(k) for its Oncopatch Surface brachytherapy system for treating localized tumors.

OncoPatch announced today that it received FDA 510(k) for its Oncopatch Surface brachytherapy system for treating localized tumors.

MiniMed (Nasdaq:MMED) announced that it received CE mark for the use of its MiniMed 780G with the Instinct sensor made by Abbott.
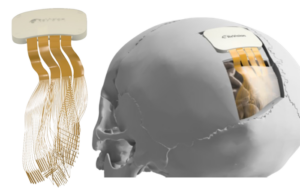
ReVision Implant announced today that it received FDA breakthrough device designation for its Occular visual cortical prosthesis.

LifeVac is approved for use as a single-use, non-powered, non-invasive airway clearance device.
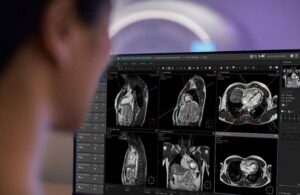
Philips (NYSE: PHG)+ announced today that it received FDA 510(k) clearance for its SmartHeart planning solution.
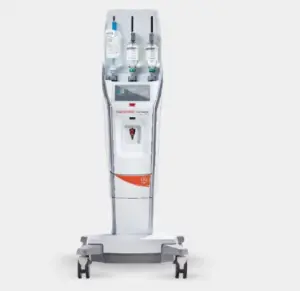
The FDA’s expanded clearances broaden the application of Bayer’s Medrad MRXperion system across a wider range of MR settings.
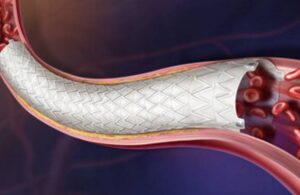
BD (NYSE: BDX)+ announced today that it received CE mark approval for its Revello vascular covered stent system.

LOS ALTOS, Calif., March 4, 2026 /PRNewswire/ — XYRA LLC announces that the US Patent and Trademark Office (USPTO) has issued a patent (No. 12,551,706) protecting the use of dose adjusted budiodarone for the treatment of atrial fibrillation (AF), with monitoring to correspondingly reduce or delay the risk of stroke and/or congestive heart failure.

TUCSON, Ariz., March 4, 2026 /PRNewswire/ — Delta Development Team, Inc., an industry leader in ruggedized thermal systems for medical and emergency applications, announced that the United States Patent and Trademark Office (USPTO) has issued U.S. Patent No. 12,566,016 covering its groundbreaking Autonomous Portable Refrigeration Unit (APRU-6L).
BROOKLYN PARK, Minn., March 4, 2026 /PRNewswire/ — Airiver Medical, a clinical stage company developing technologies to help patients who suffer from certain respiratory tract conditions, was granted designation as a Breakthrough Device from the U.S. Food and Drug Administration (FDA) for its Airiver Pulmonary Drug Coated Balloon (DCB) to treat central airway stenosis.