
Mass General Brigham works with FDA to create brain-computer interface group
The collaborators aim to resolve the clinical, regulatory, coverage and payment questions facing developers of the devices.

The collaborators aim to resolve the clinical, regulatory, coverage and payment questions facing developers of the devices.
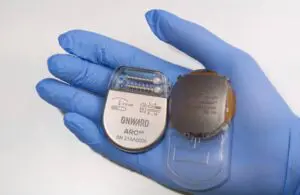
Onward Medical announced today that the FDA accepted it into its new Total Product Lifecycle Advisory Program (TAP) — a move that could boost the development of Onward’s BCI technology.

Pulse Biosciences (Nasdaq:PLSE) announced that the FDA granted 510(k) clearance for its CellFX nsPFA percutaneous electrode system.
One panelist who voted yes said the “incremental benefits outweigh the small risks of anaphylaxis.”

Getinge this week announced a major FDA clearance and the beginning of a U.S. launch of multiple surgical tools.
Lumicell announced that an FDA advisory committee voted in support of the benefit-risk profile of its LumiSight offering.

Beckman Coulter Life Sciences, a laboratory automation and innovation company, has received 510(k) clearance from the Food and Drug Administration (FDA) to distribute its DxFLEX Clinical Flow Cytometer in the United States.

SurGenTec announced today that it received FDA 510(k) clearance for its OsteoFlo HydroPutty synthetic bone graft.

Dexcom (Nasdaq:DXCM) announced today that the FDA cleared its Stelo glucose biosensor that does not require a prescription.

Medtronic (NYSE: MDT)+
announced today that it received FDA 510(k) clearance for its OsteoCool 2.0 bone tumor ablation system.