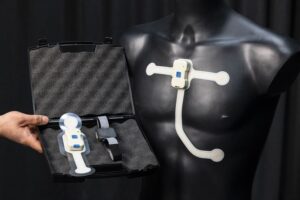
CORAL SPRINGS, Fla., Jan. 12, 2026 /PRNewswire/ — Neurolief Inc., a medical device company focused on neuromodulation therapies for neuropsychiatric conditions, today announced that the U.S. Food and Drug Administration (FDA) has approved Proliv™Rx, the first prescription, physician-directed, at-home brain neuromodulation therapy as an adjunctive treatment for adults with Major Depressive Disorder (MDD) who failed to achieve satisfactory improvement from at least one previous antidepressant medication.