QT Imaging secures FDA clearance for updated Breast Acoustic CT scanner
The scanner reconstructs 3D tomographic images by utilising both reflection-mode and transmission-mode ultrasound data.

The scanner reconstructs 3D tomographic images by utilising both reflection-mode and transmission-mode ultrasound data.
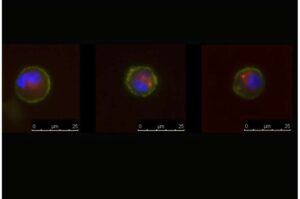
An enigmatic type of circulating tumor cell called a dual-positive (DP) cell is associated with shorter survival time in patients with advanced breast cancer, according to a study led by investigators at Weill Cornell Medicine and NewYork-Presbyterian.
Researchers from the University of California San Diego have found that a novel blood-based biomarker can predict a woman’s risk of developing dementia as many as 25 years before symptoms appear.
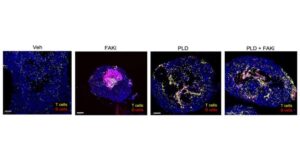
Scientists at Sanford Burnham Prebys and the University of California San Diego have published findings in Cell Reports demonstrating a treatment approach in mice that allowed more tumor-fighting cells to approach tumors.

OSLO, Norway, March 3, 2026 /PRNewswire/ — Photocure ASA (OSE: PHO), the Bladder Cancer Company, announces that its partner Asieris Pharmaceuticals (SSE: 688176) communicated today that its core product APL-1702 (trade name: CEVIRA®), has received the Drug Registration Certificate from China’s National Medical Products Administration (NMPA), enabling commercial launch in China.

TORONTO and DALLAS, March 3, 2026 /PRNewswire/ – Perimeter Medical Imaging AI, Inc. (TSXV: PINK) (OTCQX: PYNKF) (“Perimeter” or the “Company”), a commercial-stage medical technology company, announced today that it has received U.S. Food and Drug Administration (“FDA”) premarket approval (“PMA”) for Claire™ (formerly the Perimeter OCT B-Series with ImgAssist AI 2.0), the first AI-enabled imaging device approved in the United States for intraoperative breast cancer margin assessment.

DENVER, March 2, 2026 /PRNewswire/ — Ultrasound AI, a pioneer in artificial intelligence applications for medical imaging, today announced it has received FDA De Novo clearance for its flagship Delivery Date AI technology, a cloud-based SaMD that determines a Predicted Delivery Date (PDD) solely from standard ultrasound images and seamless integration into current OB/MFM prenatal visit workflows; PDD is provided in real-time for actionable decision-making by the clinical team.

A Phase I clinical trial published in The Lancet has shown that combining stem cell therapy with standard fetal surgery before birth is a safe and promising approach to treat myelomeningocele, a severe form of spina bifida.
Carea, a pregnancy and postnatal wellbeing app, is bridging this gap with the launch of its new Trying to Conceive: IVF/IUI Mode, to support the tens of thousands of women undergoing complex fertility treatment, amid growing concern over inconsistent clinic continuity and limited day-to-day guidance.
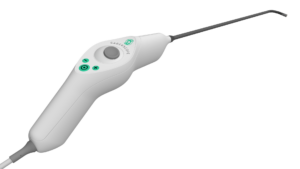
SALT LAKE CITY, Feb. 24, 2026 /PRNewswire/ — Xenocor, Inc. today announced that the U.S. Food and Drug Administration (FDA) has cleared the new Xenocor Saberscope®, a single-use 5mm articulating laparoscope designed for high definition (HD) visualization during minimally invasive abdominal (belly) and thoracic (chest) surgical procedures.