
Olympus picks up pediatric indications from FDA for duo of single-use bronchoscopes
Olympus announced today that it received FDA clearance for pediatric use for two of the single-use bronchoscopes it distributes in the U.S.

Olympus announced today that it received FDA clearance for pediatric use for two of the single-use bronchoscopes it distributes in the U.S.
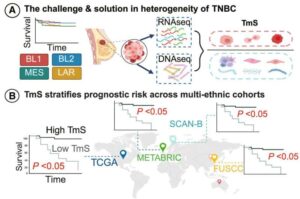
Researchers at The University of Texas MD Anderson Cancer Center have developed a new computational approach designed to better account for changes in gene expression within tumors relative to their unique microenvironments. This approach outperformed current methods for predicting chemotherapy response in patients with triple-negative breast cancer (TNBC).

Philips (NYSE: PHG)+ announced that it launched the InkSpace Imaging Snuggle pediatric body array coil for its 3.0T MRI systems.
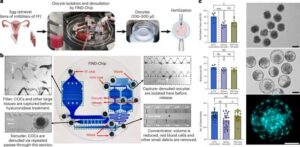
Scientists have now developed a way to find missing eggs that could bring new hope to couples seeking fertility treatment.

BRECKSVILLE, Ohio, Feb. 12, 2026 /PRNewswire/ — Applied Medical Technology, Inc. (AMT) today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for Explant Express®, a breast implant removal device designed to support efficient, controlled explantation of ruptured silicone breast implants.
With a new MRI technique that shows both heart tissue and blood flow simultaneously, physicians can see where heart defects occur and precisely plan to repair them, according to new research.
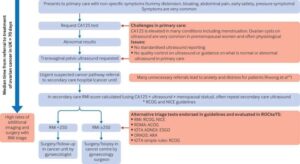
The IOTA ADNEX ultrasound tests picked up 9 out of 10 women with cancer.

An alarming 71% of grocery store baby food products in the United States are classified as ultra-processed foods (UPFs), according to new research published in the journal Nutrients.
Eyas Medical Imaging is currently scaling up operations with plans to commercialise the system in the US later this year.

More than 2,000 known CFTR mutations have been identified worldwide. The type of mutation a patient carries can alter everything from how severe their symptoms are to what drugs will work for them.