
Hair-thin fiber-optic sensors could detect cancer by reading multiple biomarkers
This breakthrough could lead to next-generation medical tools that track disease, guide treatment and monitor the body in real time

This breakthrough could lead to next-generation medical tools that track disease, guide treatment and monitor the body in real time

The method relies on the fact that deuterium, once in the body, participates in biochemical reactions alongside ordinary hydrogen and becomes incorporated into the carbon-hydrogen bonds of organic compounds.
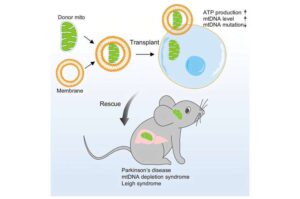
Chinese researchers have developed a novel and highly efficient mitochondrial capsule transplantation therapy.
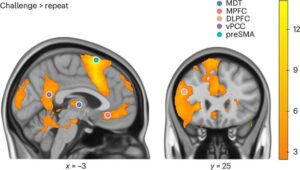
Powerful brain imaging has helped uncover why people with post-traumatic stress disorder (PTSD) who engage in negative self-talk may be struggling with the first line of treatment. The discovery, published in Nature Mental Health, sheds new light as to why underlying brain mechanisms mean some therapies potentially work for some people and not others, and could guide the development of more targeted treatments for PTSD.

Could wounded skin someday regrow perfectly without scars? A new study by Harvard stem cell biologists published in Cell reveals a way to fully regenerate skin by unblocking an embryonic healing mechanism that shuts off after birth. Demonstrated on mice, the study suggests a potential means to develop similar therapies in human patients.
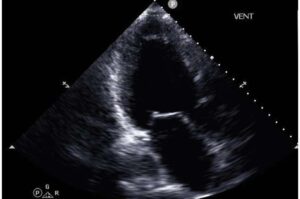
Applying artificial intelligence techniques to cardiac ultrasound data may make it easier to identify patients with advanced heart failure, a new study has found. The study—led by investigators at Weill Cornell Medicine, Cornell Tech, Cornell Ann S. Bowers College of Computing and Information Science, Columbia University Vagelos College of Physicians and Surgeons and NewYork-Presbyterian—offers the prospect of better care for many thousands of patients who may be overlooked due to the difficulty of diagnosing their condition.
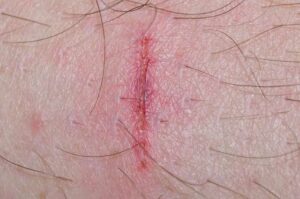
Biomedical engineers from Brown University have developed a new wound dressing material that releases antibiotic drugs only when harmful bacteria are present in a wound. In the new study, published in the journal Science Advances, the researchers show that the material could help rapidly clear wound infections to accelerate healing while reducing the unnecessary use of antibiotics—a major driver of antibiotic resistance and hard-to-treat “superbug” infections that claim tens of thousands of lives worldwide each year.
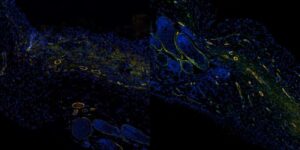
No matter the size or severity, wounds on human skin are difficult to monitor while they heal. Biopsies disrupt the wound site and are too invasive for routine, repeated monitoring, and most medical imaging devices that could do the job are large, expensive, and booked up with more pressing diagnostics. Clinicians typically resort to visual inspection or quick measurements of the wound’s size over time.

Scientists from Great Ormond Street Hospital (GOSH) and University College London (UCL) have created the first lab‑grown esophagus—the food pipe—shown to safely replace a full section of the organ and restore normal function, including swallowing, in a growing animal without the need for immunosuppression.

Researchers at Graz University of Technology (TU Graz) have developed a novel virtual reality (VR) system that could make the treatment of arachnophobia, also known as spider phobia, more targeted and personalized in the future. The work is published in the journal Frontiers in Human Neuroscience.