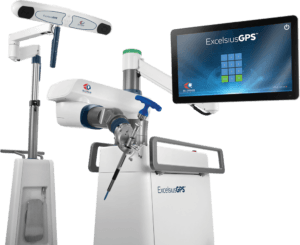
FDA clears orthopedics robot for TKA from Globus Medical
Globus Medical has won FDA clearance for its new ExcelsiusFlex orthopedics robot stereotaxic surgery in knee procedures and an expanded indication for its ExcelsiusHub system.

Globus Medical has won FDA clearance for its new ExcelsiusFlex orthopedics robot stereotaxic surgery in knee procedures and an expanded indication for its ExcelsiusHub system.

Johnson & Johnson MedTech‘s DePuy Synthes announced today that it received a new FDA 510(k) clearance for its Velys surgical robot platform.

Moon Surgical announced today that it received FDA clearance for the commercial version of its Maestro surgical robot system.

Microbot Medical (Nasdaq:MBOT) this week announced it received FDA approval to start its human clinical trial for its Liberty Endovascular Robotic Surgical System.

Undergoing surgery is seldom a pleasant experience, and it can sometimes be highly invasive. Surgical procedures have evolved steadily over the centuries, growing with the knowledge of anatomy and biology.
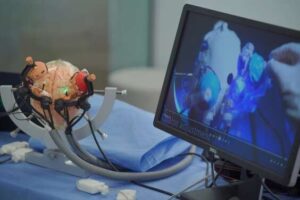
A research team has developed an interactive multi-stage robotic positioner specifically designed for magnetic resonance imaging (MRI)-guided stereotactic neurosurgery
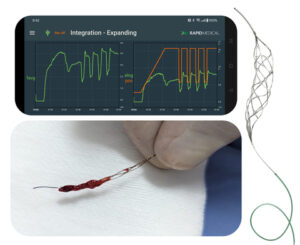
The procedures utilized an AI-powered, robotic TIGERTRIEVER to automate and optimize personalized treatments
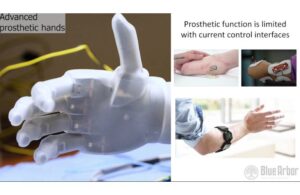
Blue Arbor Technologies announced today that it received FDA breakthrough device designation for its Restore neuromuscular interface system.

Medical Microinstruments said the system could increase the number of physicians who can perform complicated microsurgical procedures.
Near-Infrared fluorescence now available on Modus X robotic exoscope, complementing system’s existing advanced fluorescence capabilities