MedTech News
.................... by Andrew Celentano
Zepto Life Technology’s mould infection panel obtains FDA breakthrough designation
Zepto Life’s FungiFlex mould panel is designed to identify 14 clinically important mould species based on cell free fungal DNA detected in plasma.

Tandem wins CE mark for automated insulin delivery in type 2 diabetes, pregnancy
Tandem Diabetes Care (Nasdaq:TNDM) announced today that it received expanded CE mark for its automated insulin delivery (AID) systems.
Lab-Grown Brains Could One Day Help Reawaken Nerve Regeneration
Lab-grown brains could help scientists reawaken nerve regeneration.
GE HealthCare earns FDA nod for AI-enabled radiotherapy tech
GE HealthCare (Nasdaq:GEHC) announced today that it received FDA 510(k) clearance for its MIM Contour ProtégéAI+ 2.0 software.
A new vaccine adjuvant could make it easier to eradicate polio
The adjuvant can help the injectable polio vaccine induce a strong immune response in the GI tract, which is considered critical to eradicating the virus.
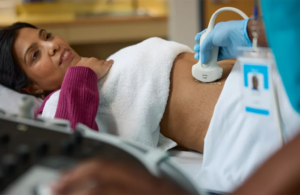
Philips wins FDA clearance for AI-powered ultrasound workflow tech
Philips (NYSE:PHG) announced today that it received FDA clearance for its Elevate Plus advanced imaging and AI and automation capabilities.
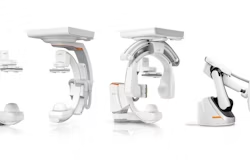
Pulmera wins FDA clearance for 3D imaging platform
Pulmera announced today that it received FDA 510(k) clearance for its CBeam 3D intraoperative imaging platform.

Edwards gets FDA nod for Triformis Resilia surgical tricuspid valve replacement
Edwards Lifesciences (NYSE:EW) said today that it received FDA approval for Triformis Resilia, its surgical tricuspid valve replacement.
