MedTech News
.................... by Andrew Celentano
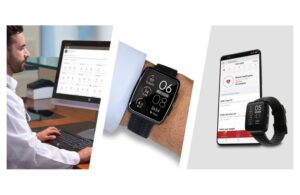
FDA clears Masimo W1 medical watch for telemonitoring connectivity
Masimo (Nasdaq: MASI)+
announced today that the FDA granted 510(k) clearance for its W1 medical watch for connectivity purposes

Walking the walk, researchers develop motion compatible brain scanner
Researchers tested their prototype in a real world setting to assess its accuracy and determine needs for improvement.
Invenio Imaging Announces First Patients Enrolled in US Pivotal Study of AI-Based Image Analysis Module for Lung Cancer
SANTA CLARA, Calif., Aug. 9, 2024 /PRNewswire/ — Invenio Imaging, a leader in intraoperative fresh tissue imaging and artificial intelligence (AI), announced today the enrollment of the first patients in a US pivotal study of its AI-based image analysis module for lung cancer

Genova Diagnostics Partners with Neurocode to Launch Innovative Alzheimer’s Assessment
ASHEVILLE, N.C., Aug. 9, 2024 /PRNewswire/ — Genova Diagnostics, a global leader in clinical laboratory services, proudly announces the launch of its groundbreaking Alzheimer’s Assessment, a non-invasive blood test utilizing the p-Tau217 biomarker for early detection of Alzheimer’s disease. This initiative is enabled by a strategic partnership with Neurocode, a leading innovator in neurological diagnostics and research.

Innovative Anti-Nausea Smartwatch EmeTerm® Smart Set to Launch on Indiegogo
VANCOUVER, BC, Aug. 8, 2024 /PRNewswire/ — EmeTerm® Smart uses patented hybrid technology to seamlessly integrate the functionality of a smartwatch with that of an anti-nausea wristband, ensuring a carefree and healthy life for all. EmeTerm® Smart is launching on Indiegogo as the world’s first anti-nausea smart watch.
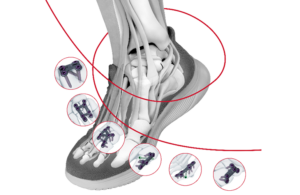
J&J’s DePuy Synthes launches new plating system
J&J MedTech’s (NYSE: JNJ)+
DePuy Synthes unit today announced that it launched its TriLeap lower extremity anatomic plating system.

Pulse Biosciences touts first-in-human pulsed field ablation cases
Pulse Biosciences (Nasdaq:PLSE) today announced the first completed cases in a first-in-human feasibility study of its pulsed field ablation system.
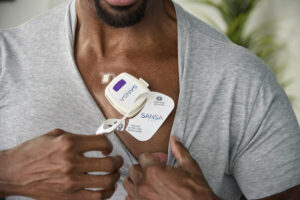
U.S. Food and Drug Administration Gives Huxley Medical 510(k) Clearance for SANSA Home Sleep Apnea Test
ATLANTA, Aug. 7, 2024 /PRNewswire/ — Huxley Medical, a developer of technologies that streamline cardiopulmonary care, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its groundbreaking, chest-worn, sleep apnea diagnostic patch, SANSA.
