MedTech News
.................... by Andrew Celentano
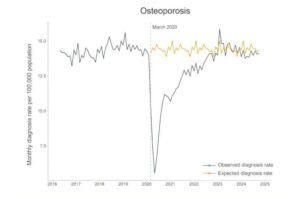
The pandemic’s hidden toll: Millions of chronic conditions left undiagnosed
When COVID hit, health care systems around the world were turned upside down. Hospitals cleared beds, routine appointments were canceled and people were told to stay at home unless it was urgent.
Single gene found to influence gut bacteria balance and IBD susceptibility
Two recent studies from the University of California, Riverside, published in the same issue of Gut Microbes highlight the role of a gene called PTPN2 in protecting the gut from harmful bacteria linked to inflammatory bowel disease (IBD).

Using lab-grown lung tumors as test subjects for tailored cancer therapies
Lung cancer varies widely from patient to patient, and that diversity makes it hard to find effective treatments. Researchers at the Berlin Institute of Health at Charité (BIH) have developed a method to evaluate multiple therapeutic approaches on patient-derived “tumoroids”—miniature tumors grown from tissue removed during surgery at Charité.

Affera™ momentum continues as Medtronic announces CE Mark in Europe and U.S. IDE first cases for Sphere-360™ PFA catheter to treat paroxysmal atrial fibrillation
First-of-its-kind, rotation-free single-shot PFA catheter supported by strong safety and efficacy data adds to the groundbreaking Affera family of technologies in Europe

Cancer’s secret safety net
Researchers uncover a hidden mechanism that allows cancer to develop aggressive mutations.
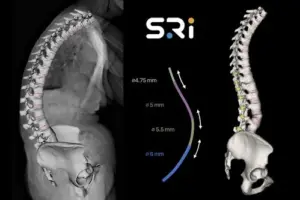
Spinal Resources Strengthens IP Portfolio with New Bezier Rod Patent
The patent’s focus on a constant-slope transition ensures a gradual and predictable stiffness profile
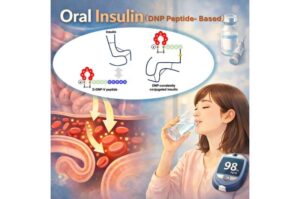
Advancing the realization of oral insulin using novel peptide technology
For more than a century, oral insulin has been considered a “dream” therapy for diabetes, hindered by enzymatic degradation in the digestive tract and the absence of a dedicated intestinal transport mechanism. Consequently, many patients must rely on daily insulin injections, which can significantly reduce their quality of life.
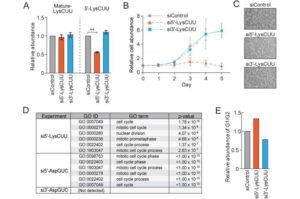
Tiny molecules called tRNA halves may contribute to prostate cancer cell growth
Prostate cancer is the second-most common cancer in men. A new study from Thomas Jefferson University uncovered a new potential therapeutic target in tiny molecules called tRNA halves.
