MedTech News
.................... by Andrew Celentano

An injectable particle could make surgery safer for infants
Biomedical researchers have designed an injectable microgel to help reduce bleeding in infants who require surgical care. In an animal model, the engineered microgel reduced bleeding by at least 50%. The paper, “Hemostatic B-Knob Triggered MicroGels (BK-TriGs) to Address Bleeding in Neonates,” is published in the journal Science Advances.

Supercomputer simulations reveal early red blood cell damage in blood pumps
For patients with heart failure, blood pumps can be lifesaving. But the very forces that sustain circulation can also harm it, damaging red blood cells through hemolysis and compromising the body’s oxygen supply. Now, supercomputer simulations are revealing how red blood cells deform under stress, offering new insights that could lead to safer, more effective blood pump designs.

Scalable sensors lower the cost of studying genetic disorders
Researchers have demonstrated a new class of low-cost, scalable sensors that can be used to monitor electrical activity in human cerebral organoids. Because electrical signals are key to understanding brain function, this advancement facilitates research into both neurodevelopment and genetic disorders such as Angelman syndrome.
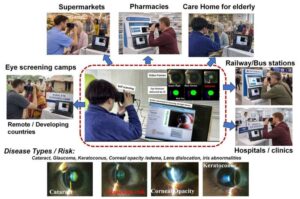
AI-powered portable eye scanner expands access to low-cost community screening
Imagine being able to assess how healthy the front of our eyes are not only in hospitals, but also in remote eye-screening camps, elderly-care facilities, pharmacies, or even train stations. That is the future a research team led by Professor Toru Nakazawa at the Graduate School of Medicine, Tohoku University is working toward with a newly developed portable AI-powered scanning slit-light device. This convenient device hopes to make ophthalmic care more accessible, so patients can be assessed any place and any time. The findings are published in Scientific Reports.
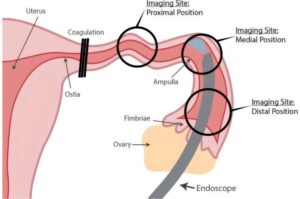
A redesigned endoscope offers a new way to look for early signs of ovarian cancer
Ovarian cancer remains the deadliest gynecologic cancer, largely because it is rarely found early. Symptoms are often vague, and existing screening approaches—such as blood tests and transvaginal ultrasound—can miss the disease at stages when treatment is most effective. In recent years, research has reshaped understanding of how many aggressive ovarian cancers begin, pointing not to the ovary itself, but to the fallopian tubes. That shift has created a need for tools that can safely examine these narrow structures for early changes linked to cancer.

Transcranial magnetic stimulation can target a deep brain region without surgery or medication
Neuroscientists at University of Iowa Health Care have demonstrated for the first time that noninvasive brain stimulation can alter the activity of a critical deep brain region involved in emotion and memory. Moreover, the study shows that personalizing the stimulation site using a patient’s unique brain connectivity pathway can increase the neuromodulation effect.

Zeto Announces FDA 510(k) Clearance of Zeto New Wave™, Advancing Efficiency in Outpatient EEG
RALEIGH, N.C., April 2, 2026 /PRNewswire/ — Zeto, Inc., a medical technology company dedicated to making brain health insights through EEG a foundational vital sign in healthcare, announced that it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for Zeto New Wave™, its newest EEG system for outpatient clinics and homes.
MediBeacon secures CE Mark certification for TGFR monitor and sensor
Lumitrace is currently approved in China and the US, with submission to EU regulatory authorities pending.
