MedTech News
.................... by Andrew Celentano
Intuitive wins FDA clearances for cardiac procedures on da Vinci 5
Intuitive Surgical (Nasdaq: ISRG)+
has received FDA clearance for “several cardiac procedures,” CEO Dave Rosa said today while announcing the surgical robotics company’s latest financial and operating results.
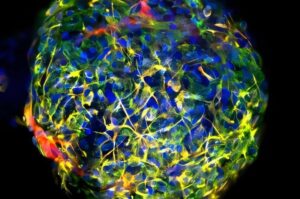
Automated 3D heart tissue platform speeds up cardiac drug testing
Researchers from Leiden University Medical Center (LUMC) and the Leiden-based biotech company Ncardia have joined forces to develop a new platform that enables the automated production of 3D cardiac microtissues.
Medical-grade near-infrared light therapy shows promise for protecting football players’ brains
Punch-drunk syndrome, boxer’s madness, chronic traumatic encephalopathy (CTE). The name has changed over the years, but the cause is clear: repeated impacts can affect long-term brain health, with symptoms ranging from confusion to memory loss and potentially dementia.
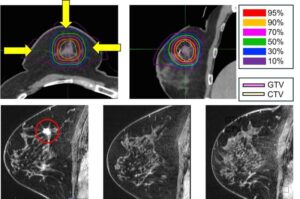
Carbon-ion therapy offers nonsurgical option for early breast cancer treatment
For many women with early breast cancer, surgery is effective but life-altering. New five-year data from the National Institutes for Quantum Science and Technology (QST) suggest that a precisely targeted, high-energy particle therapy may allow some patients to avoid surgery without compromising oncologic outcomes.

How pancreatic tumors use the MYC protein to evade immune detection
An international research team has succeeded in deciphering a key mechanism that controls the growth of pancreatic cancers.
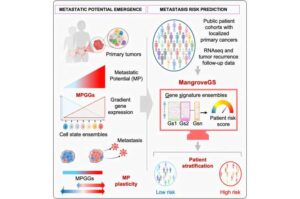
AI tool predicts cancer metastasis risk using gene expression signatures
Why do some tumors spread while others remain localized? The mechanisms governing the metastatic potential of tumor cells remain largely unknown—yet understanding this is crucial for optimizing patient care.
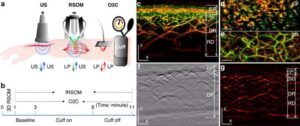
Skin imaging scan can detect early signs of heart disease
Researchers from Helmholtz Munich and the Technical University of Munich (TUM) have developed “fast-RSOM,” a new imaging technology that can capture detailed images of the smallest blood vessels directly through the skin—without the need for invasive procedures.

Aidoc Secures FDA Clearance for Healthcare’s First Comprehensive Foundation Model AI
11 newly cleared indications, combined with three existing ones, introduce an AI safety net for crowded Emergency Departments and imaging backlogs
