MedTech News
.................... by Andrew Celentano
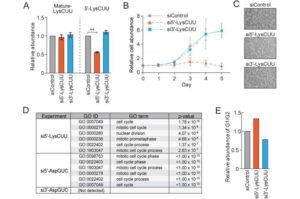
Tiny molecules called tRNA halves may contribute to prostate cancer cell growth
Prostate cancer is the second-most common cancer in men. A new study from Thomas Jefferson University uncovered a new potential therapeutic target in tiny molecules called tRNA halves.
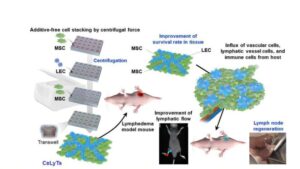
Regenerating lost lymph nodes with bioengineered tissues
The rising incidence of cancer worldwide has led to an increasing number of surgeries that involve the removal of lymph nodes. Although these procedures play a major role in cancer staging and preventing the spread of malignancies, they sometimes come with severe long-term consequences.
Microbes living in our mouths could hold the key to obesity prevention
Scientists may have found a new way to spot early signs of obesity, which could lead to novel prevention strategies. A study published in the journal Cell Reports has discovered that people living with obesity have a distinct set of microbes in their mouths compared to people at a healthy weight.
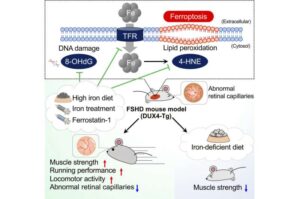
Iron supplements restore muscle strength in mouse model of muscular dystrophy
Researchers at Kumamoto University have demonstrated that iron supplementation can significantly alleviate muscle pathology and functional decline in a mouse model of facioscapulohumeral muscular dystrophy (FSHD), a rare genetic muscle disease for which no effective treatment currently exists.

Woori IO, an OSR Company, Highlights FDA Clarification on Non-Medical Wearables as a Catalyst for Accelerated Commercial Launch
BELLEVUE, Wash., Jan. 22, 2026 /PRNewswire/ — Woori IO, an OSR company developing next-generation noninvasive glucose monitoring technologies, today commented on the U.S. Food and Drug Administration’s recent clarification regarding non-medical-grade wearable devices, noting that the FDA’s position establishes a regulatory framework highly conducive to Woori IO’s commercialization strategy.

Your Breath May Reveal Hidden Changes in the Gut Microbiome
Learn how chemicals released by gut microbes end up in exhaled breath, and how researchers used those signals to identify microbial communities linked to disease.
Zimmer Biomet launches new foot and ankle system
Zimmer Biomet (NYSE: ZBH)+
recently announced the Brachiator mini-rail external fixation system.

Bridge to Life wins FDA De Novo for transplant liver perfusion system
Bridge to Life has received FDA De Novo clearance for its VitaSmart hypothermic oxygenated perfusion (HOPE) system for liver transplantation.
