MedTech News
.................... by Andrew Celentano

A ‘window to the brain’: Chip tracks glioblastoma treatment response using tumor vesicles in blood
Technology created at the University of Queensland could improve the odds of surviving brain cancer and change how we treat a range of neurological conditions.

A portable ultrasound sensor may enable earlier detection of breast cancer
For people who are at high risk of developing breast cancer, frequent screenings with ultrasound can help detect tumors early.

Pink noise reduces REM sleep and may harm sleep quality
Pink noise—often used to promote sleep—may reduce restorative REM sleep and interfere with sleep recovery. In contrast, earplugs were found to be significantly more effective in protecting sleep against traffic noise.

Experimental immunotherapy clears harmful artery cells, reducing plaque in mice
Scientists have designed an immunotherapy that reduces plaque in the arteries of mice, presenting a possible new treatment strategy against heart disease.

Anticipating aging-related mental decline using saliva samples and AI
As humans age beyond early adulthood, their physical and mental functions tend to slowly worsen over time. One of the most common sources of severe mental decline in older adults are neurodegenerative diseases, conditions characterized by the progressive loss of neurons in the brain or peripheral nervous system.
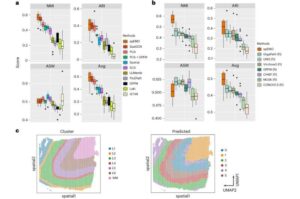
New AI tool helps scientists see how cells work together inside diseased tissue
A new study from Yale University researchers shows how artificial intelligence can bring image, gene and protein data together, offering a clearer picture of what is happening inside the body and how diseases develop.
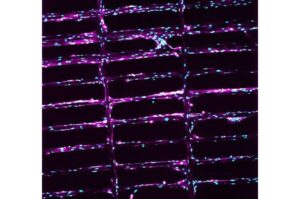
Engineered micro scaffolds show promise for helping people recover from severe muscle loss
When a car accident or athletic injury destroys more than 20% of a muscle’s mass, the body faces a problem it often can’t heal fully on its own. Without intervention, scar tissue fills the injury site and can leave patients with permanent weakness and limited mobility.

Researchers discover genetic ancestry is a critical component of assessing head and neck cancerous tumors
Genetic ancestry plays a key role in determining the behavior of head and neck tumors and may help explain why African-American patients survive for half as long as their counterparts of European ancestry, according to a new review study led by researchers from the University of Maryland School of Medicine’s (UMSOM) Institute for Genome Sciences (IGS) and the University of Maryland Greenebaum Comprehensive Cancer Center (UMGCCC).
