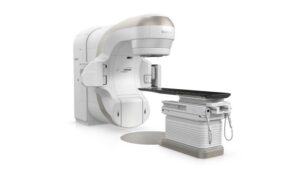
FDA clears Varian’s TrueBeam systems for low-dose radiation therapy in osteoarthritis
Growing evidence has demonstrated that low-dose radiation can target inflammation and reduce pain for osteoarthritis patients.

Growing evidence has demonstrated that low-dose radiation can target inflammation and reduce pain for osteoarthritis patients.
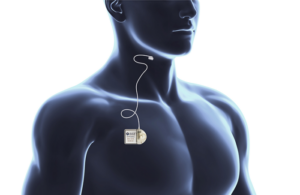
LivaNova (Nasdaq: LIVN)+ announced today that it received FDA premarket approval (PMA) for its aura6000 neuromodulation system for sleep apnea.

JenaValve announced today that it received FDA premarket approval (PMA) for its Trilogy THV system.

Nia Therapeutics announced today that it received FDA breakthrough device designation for its Smart Neurostimulation System (SNS).

SUZHOU, China, March 18, 2026 /PRNewswire/ — Peijia Medical (9996.HK), a leading Chinese domestic company in the high-growth transcatheter valve therapeutics and neurovascular interventions markets, announced that its DCwire® Micro Guidewire has received 510(k) clearance from the U.S. Food and Drug Administration (“FDA”) on March 17, 2026. The product was independently designed and developed by Achieva Medical Limited, a subsidiary of the Company.
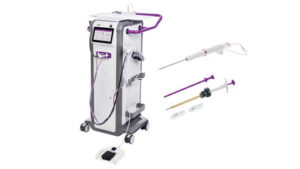
CINCINNATI, March 18, 2026 /PRNewswire/ — Mammotome, a Danaher company, announces FDA Clearance of two innovations for MR-guided breast biopsy: the Mammotome Prima™ MR Dual Vacuum-Assisted Breast Biopsy System – the industry’s first solution designed to be positioned in the MRI scanner room at the patient’s side – and the HydroMARK™ Plus Breast Biopsy Site Marker for MR, engineered for exclusive use with the Mammotome Prima™ system universal targeting set. Following a successful debut of the Mammotome Prima™ MR system in Europe, both products will be introduced to U.S. clinicians at the 2026 Society of Breast Imaging (SBI) Symposium. By bringing the device directly into the scanner room, the Mammotome Prima™ MR system enhances clinician coordination, streamlines communication, and helps maintain consistent focus on the patient throughout the procedure.

CARLSBAD, Calif., March 18, 2026 /PRNewswire/ — Spinal Elements®, a spine technology company known for developing innovative surgical solutions that prioritize clinical performance and surgical efficiency, today announced 510(k) clearance from the FDA and the first cases using its Ventana A Anterior Lumbar Interbody Fusion (ALIF) System. Ventana A strengthens Spinal Elements’ family of 3D printed titanium interbodies for lumbar and cervical fusion and is being introduced through a limited market launch.

MiniMed Flex™ is about half the size of the previous generation MiniMed™ 780G system, and features the SmartGuard™ adaptive algorithm to automatically adjust and autocorrect insulin delivery in real-time

Prodeon Medical announced today that it received FDA 510(k) clearance for its Urocross expander system.

Revolve Surgical announced today that it received FDA 510(k) clearance for its surgical robot platform.