
FDA clears computer-assisted thrombectomy system from Penumbra
Penumbra (NYSE:PEN) announced today that it received FDA clearance for and began the launch of its Lightning Flash 2.0 CAVT system.

Penumbra (NYSE:PEN) announced today that it received FDA clearance for and began the launch of its Lightning Flash 2.0 CAVT system.

PLEASANTON, Calif., April 22, 2024 /PRNewswire/ — Expanse ICE announced today the ICE Aspiration System has received 510(k) clearance from the U.S. Food and Drug Administration. This announcement introduces a new and exciting player in the peripheral thrombectomy market.
Philips (NYSE: PHG)+
announced today that the FDA granted 510(k) clearance for its Zenition 30 mobile C-arm.
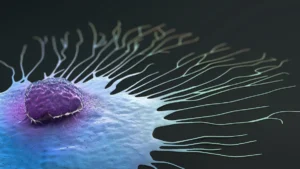
Lumicell developed the technology to help physicians detect residual cancer in the breast cavity after surgery.

The Osia System, which launched commercially in 2020, is indicated for people with conductive hearing loss, mixed hearing loss and single-sided sensorineural deafness (SSD)

Lumicell today announced it received FDA approval for its LumiSystem direct visualization system for breast cancer removal.

The company’s 3rd FDA clearance accelerates the shift to AI-powered digital hematopathology, enhancing diagnostic speed and patient care

IRVING, Texas, April 16, 2024 /PRNewswire/ — Xstim, Inc., a pioneering developer and manufacturer of cutting-edge bone growth stimulation systems, is thrilled to announce its recent Premarket Application (PMA) approval from the U.S. Food and Drug Administration (FDA) for Xstim™ Spine Fusion Stimulator

The 3D printed PEEK implants using the EXT 220 MED were successfully demonstrated in nearly 40 cranioplasties across Europe in recent months says the company.
Simpson Interventions announced this week that it received FDA breakthrough designation for its Acolyte image-guided crossing and re-entry catheter system.