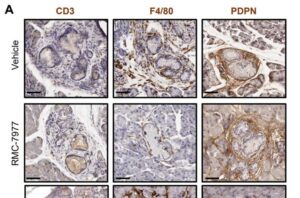
New strategy intercepts pancreatic cancer by eliminating microscopic lesions before they become cancer
A new preclinical study in mice shows that precancerous cells in the pancreas can be eliminated before they have the chance to become tumors.

A new preclinical study in mice shows that precancerous cells in the pancreas can be eliminated before they have the chance to become tumors.
Now researchers in the former Laboratory of Chromatin Biology and Epigenetics, headed by C. David Allis until his death in early 2023, have discovered surprising new details about the mechanics of this protein complex, which could potentially lead to new cancer therapies. The findings were published in Genes & Development.
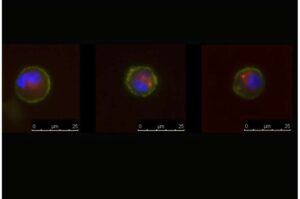
An enigmatic type of circulating tumor cell called a dual-positive (DP) cell is associated with shorter survival time in patients with advanced breast cancer, according to a study led by investigators at Weill Cornell Medicine and NewYork-Presbyterian.

View offers 2D and 3D visualisation, along with AI-based tools, to assist in clinical decision making.

MiniMed (Nasdaq:MMED) announced that it received CE mark for the use of its MiniMed 780G with the Instinct sensor made by Abbott.

Matthew Jones is using experimental technology and computation to decode the molecular processes that underlie the way tumors evolve to resist treatment by pinpointing how and when aggressive mutations arise.
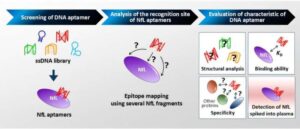
Leveraging the potential of NfL as a biomarker, researchers from Japan have developed the world’s first aptamer (tiny, single-stranded synthetic DNA molecules) that can bind NfL with high affinity and specificity

Researchers at the Indiana University Melvin and Bren Simon Comprehensive Cancer Center have found that depleting a clotting protein made by the liver could slow down pancreatic cancer.
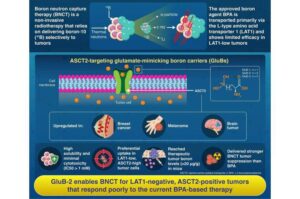
Boron agents termed GluBs, developed by Science Tokyo researchers, have overcome a key limitation in cancer therapy by entering tumor cells through a pathway that standard drugs cannot use.
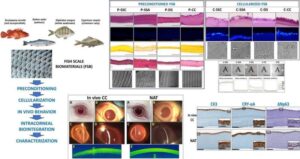
Now, researchers from the Tissue Engineering Group of the Department of Histology at the University of Granada’s Faculty of Medicine and the ibs.GRANADA Institute for Biomedical Research have created highly biocompatible, resistant, and transparent corneal implants from scales of various types of fish commonly found on the market, such as carp. The findings are published in the journal Materials & Design.