Overture Orthopaedics Announces Milestone $1.0Mn in Sales of its OvertureTi Knee Resurfacing System
OvertureTi Knee Resurfacing System implants are designed specifically as an alternative when it is still too early for arthroplasty

OvertureTi Knee Resurfacing System implants are designed specifically as an alternative when it is still too early for arthroplasty

Scientists at Johns Hopkins Medicine say results of a new study are advancing efforts to exploit a new target for Alzheimer’s disease: a protein that manufactures an important gas in the brain.

As the year comes to a close, it’s time to look back at the numerous innovations across the diabetes technology industry in 2025.

CardioKG provides a detailed view of the heart’s structure and function which dramatically improves the accuracy of predicting which genes are linked to disease and whether existing drugs could treat them.
A research team at Oregon Health & Science University has discovered a promising new drug combination that may help people with acute myeloid leukemia overcome resistance to one of the most common frontline therapies.
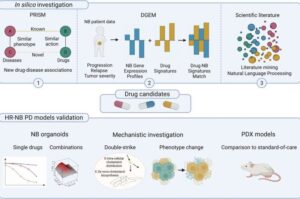
Researchers at Lund University in Sweden have identified a combination of statins and phenothiazines that is particularly promising in the treatment of the aggressive form of neuroblastoma.

Texas A&M University researchers have found that a brief virtual reality (VR) simulation of a near-death experience can sharply reduce people’s anxiety about death. In a small pilot study of about 60 young adults, participants reported a 75% decrease in their fear of death after a single 12-minute session.
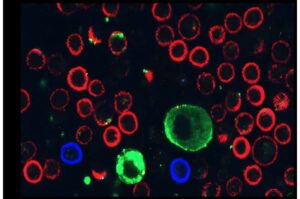
A new international study led by Prof. Carmit Levy of the Department of Human Genetics and Biochemistry at the Gray Faculty of Medical & Health Sciences at Tel Aviv University finds that melanoma cancer cells paralyze immune cells by secreting extracellular vesicles (EVs).
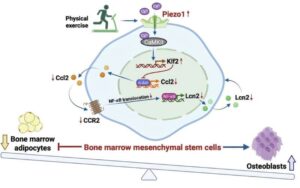
A research team from the Department of Medicine, School of Clinical Medicine, LKS Faculty of Medicine at the University of Hong Kong (HKUMed) has uncovered a key biological mechanism that explains how exercise maintains strong bones.
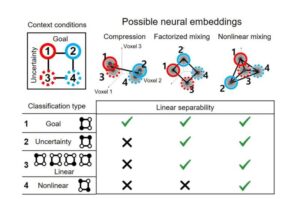
The research team led by Professor Sang Wan Lee from the Department of Brain and Cognitive Sciences, in collaboration with IBM AI Research, has deciphered how the human brain manages goal changes in uncertain situations.