
New tool launches to support women through post-loss journey
https://med-techinsights.com/2026/04/22/new-tool-launches-to-support-women-through-post-loss-journey/

https://med-techinsights.com/2026/04/22/new-tool-launches-to-support-women-through-post-loss-journey/
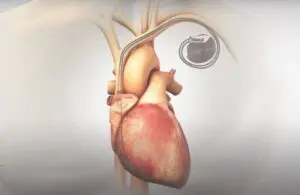
Orchestra BioMed (Nasdaq:OBIO) announced today that it received a second FDA breakthrough device designation for its AVIM therapy.
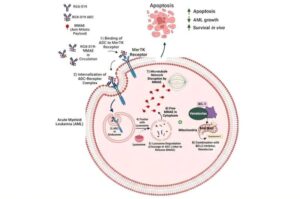
New research out of VCU Massey Comprehensive Cancer Center has uncovered a targeted cancer therapy that significantly prevents leukemia progression, improves survival rates and minimizes damage to surrounding healthy tissue.
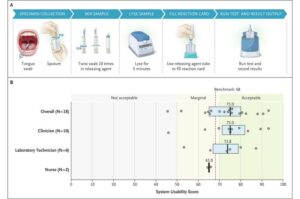
Led by researchers from Heidelberg Faculty of Medicine at Heidelberg University and the University of California, San Francisco (U.S.), an international team has evaluated a novel approach for the diagnosis of tuberculosis.
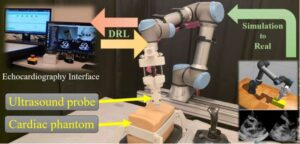
A Concordia-led team of researchers has developed a new AI-driven robotic system that can perform cardiac ultrasound scans autonomously. The researchers say this approach could expand access to cardiac imaging in remote or underserved areas, reduce operator fatigue, and standardize scan quality. The work is published in the journal IEEE Transactions on Medical Robotics and Bionics.
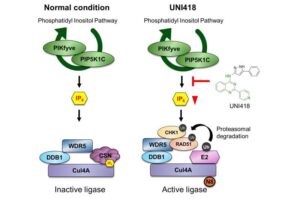
A research team led by Director Myung Kyungjae at the Center for Genomic Integrity within the Institute for Basic Science (IBS), in collaboration with Lee Joo-Yong (Chungnam University) has now uncovered a new strategy to overcome this resistance. T

GRAND RAPIDS, Mich., April 30, 2026 /PRNewswire/ — Capitan Orthopedics, Inc. (https://capitanortho.com/) announced today that it has been granted Breakthrough Device Designation from the U.S. Food & Drug Administration (FDA) for its SupraSpacerTM implant.

FDA authorization marks critical milestone enabling advancement of next-generation bone regeneration therapy for patients with degenerative spinal diseases
Avatar’s software translates CT and MR imaging data into 3D images to streamline surgical planning and operating room workflows.
Researchers at McGill University have developed a rapid way to engineer blood clots that stop severe bleeding and support tissue healing more effectively.