MedTech News
.................... by Andrew Celentano
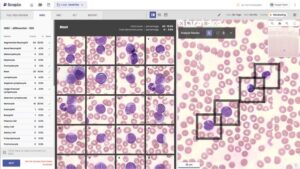
Scopio Labs Achieves EU IVDR Certification for AI-Powered Full-Field Digital Morphology Platforms
PARSIPPANY, N.J., Feb. 25, 2026 /PRNewswire/ — Scopio Labs today announced it has achieved IVDR certification from BSI, a major regulatory milestone that clears the path for its AI-driven digital morphology platforms in the European Union
Polaroid Therapeutics (PTx) Receives CE Mark for POLTX_Fiber™: the first application of APT™ to launch a new standard in wound care
ZURICH, Feb. 25, 2026 /PRNewswire/ — Polaroid Therapeutics (PTx) today announces that POLTX_Fiber™ has received the CE Mark as a Class IIb medical device with APT™ (Antimicrobial Polymer Technology).

4WEB Medical Receives 510(k) Clearance to Market its New SI Joint Truss System™
DALLAS, Feb. 25, 2026 /PRNewswire/ — 4WEB Medical, an orthopedic implant company focused on developing innovative implants that utilize its proprietary TRUSS Implant Technology™, announced that it has received 510(k) clearance to market its SI Joint Truss System™.
New blood test signals who is most likely to live longer, study finds
As people age, it becomes harder to know who is on track for healthy years ahead and who may be at higher risk for serious decline. A new study suggests that part of the answer may already be circulating in the bloodstream.
Cancer‑Eating Bacteria Could Devour Tumors From the Inside Out
Learn how reseachers are working to engineer certain bacteria to consume tumors.

Medtronic launches MiniMed Go smart MDI system with Simplera sensor in Europe
Medtronic (NYSE:MDT) announced today that it launched its MiniMed Go smart multiple daily injection (MDI) system with the Simplera sensor.
Carea launches support platform for complex fertility treatment
Carea, a pregnancy and postnatal wellbeing app, is bridging this gap with the launch of its new Trying to Conceive: IVF/IUI Mode, to support the tens of thousands of women undergoing complex fertility treatment, amid growing concern over inconsistent clinic continuity and limited day-to-day guidance.
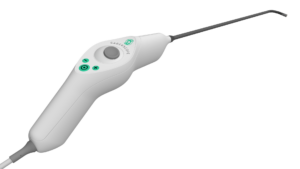
Xenocor Announces FDA Clearance of Saberscope®, the First Single-Use 5mm Articulating Laparoscope for Enhanced Surgical Visualization
SALT LAKE CITY, Feb. 24, 2026 /PRNewswire/ — Xenocor, Inc. today announced that the U.S. Food and Drug Administration (FDA) has cleared the new Xenocor Saberscope®, a single-use 5mm articulating laparoscope designed for high definition (HD) visualization during minimally invasive abdominal (belly) and thoracic (chest) surgical procedures.
