MedTech News
.................... by Andrew Celentano

Implanted nerve stimulation for obstructive sleep apnea found to be safe and effective
Researchers conducted the OSPREY (treating Obstructive Sleep aPnea using taRgEted hYpoglossal nerve stimulation) trial to better understand the safety and efficacy of pHGNS.
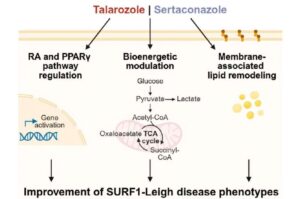
AI screening in brain organoids uncovers two drug candidates for Leigh syndrome
Together with a team from the University of Luxembourg, researchers from Heinrich Heine University Düsseldorf (HHU) and University Hospital Düsseldorf (UKD) have succeeded in utilizing artificial intelligence (AI) to establish a model that enables a better understanding of Leigh syndrome.
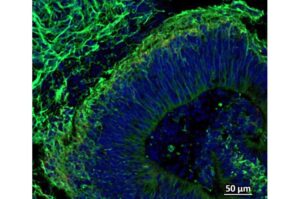
Mini brain-like structures grown in lab may help scientists treat, diagnose and stage Alzheimer’s disease
Scientists from Johns Hopkins Medicine report new evidence that clusters of brain tissue derived from the cells of patients with Alzheimer’s disease may be used to evaluate how certain patients with the neurodegenerative condition may respond to drugs commonly prescribed to treat psychiatric symptoms of the disorder.
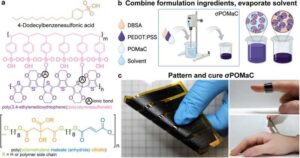
A new skin-hugging heart monitor material could make long-term ECG tracking far more comfortable
Researchers have created heart monitoring sensors that conform to the skin, are comfortable, and can be worn while people are moving.

BVI wins CE mark for Virtuoso dual-function platform
BVI Medical announced today that it received CE mark for its Virtuoso next-generation phaco-vitrectomy surgical platform.
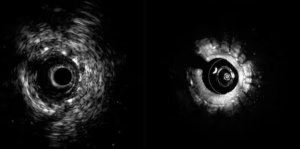
Conavi wins FDA clearance for intravascular ultrasound platform
Conavi Medical announced today that it received FDA 510(k) clearance for its next-generation hybrid imaging system.

Labcorp Launches FDA-Cleared Fentanyl Test Delivering Results in 10 Minutes
Fentanyl Urine Visual Test is available through Labcorp Point of Care, previously known as MEDTOX Diagnostics
Robust flu protection may rely on B cells that are long-lived residents in the lungs
By identifying the molecular mechanisms that control BRM cell formation, the new study provides a clearer paradigm for understanding how long-term immunity is established in the respiratory tract.
