MedTech News
.................... by Andrew Celentano
Artera’s prostate biopsy and breast cancer assays receive CE mark
The ArteraAI prostate biopsy assay predicts the risk of prostate cancer progression.
Xeltis gains EU CE mark on positive trial data for vascular access graft
Approval for Xeltis’s aXess device was based on pivotal trial data that demonstrated 79% secondary patency and a patency related reintervention rate of 1.3 per patient year.
Johnson & Johnson wins CE mark for new Ethicon 4000 surgical stapler
Johnson & Johnson (NYSE:JNJ) announced today that it received CE mark approval for its new Ethicon 4000 surgical stapler.
Philips gets FDA green light for scanner platform
Philips (NYSE:PHG) announced today that it received FDA 510(k) clearance for its Rembra platform of scanning systems.
Sonorous Neurovascular earns FDA clearance for next-gen neurovascular catheter
Sonorous Neurovascular announced today that it received FDA 510(k) clearance for its BosCATH next-generation neurovascular catheter.

Luminoah wins FDA clearance for enteral feeding pump system
Luminoah announced today that it received FDA clearance for Luminoah Flow, an enteral feeding system for those who rely on tube feeding.
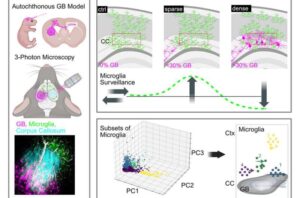
Advanced imaging uncovers immune cells’ changing role during glioblastoma invasion
Researchers from DZNE, University Hospital Bonn and the Cluster of Excellence ImmunoSensation at the University of Bonn have captured this infiltration process in the living brain with advanced microscopy.
Single-cell tool predicts cancer survival by pinpointing harmful tumor cells
Oregon Health & Science University researchers have developed a first-of-its-kind method to predict cancer patient survival using advanced molecular data from individual cells.
