MedTech News
.................... by Andrew Celentano
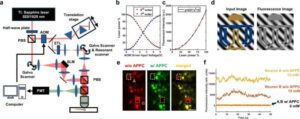
Smart laser dimmer cuts neural crosstalk in brain-circuit imaging and control
Functioning as a smart dimmer, it can selectively adjust the brightness of every pixel during laser scanning, and prevent unintended neural activation, thereby significantly enhancing the precision of all-optical brain manipulation and imaging.

Biosensor works with blood samples to identify pancreatic cancer in early stages
Brazilian researchers have developed an electrochemical sensor that can detect pancreatic cancer in its early stages.

New tool can see how different brain cell types work together
Researchers from Boston University’s Chobanian & Avedisian School of Medicine, College of Arts & Sciences, College of Engineering and Faculty of Computing & Data Sciences have developed a tool called PhysMAP to separate the “voices” of individual cell types within a crowd of electrical noise by combining several complementary features of each type’s electrical signature.
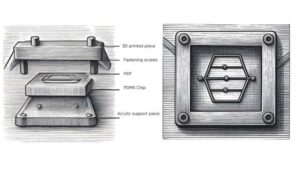
Microfluidic chip reveals how living glioblastoma slices resist chemotherapy
Combining microchip engineering techniques with cutting-edge gene profiling, scientists at Columbia University have developed a new way to study drug responses in living slices of human brain tumor cells.

Deep brain ultrasound uncovers pain-processing network tied to heat sensitivity changes
A new noninvasive neurostimulation technique capable of reaching deep regions of the brain has been used to elucidate the brain’s pain mechanisms, with promising clinical applications in neurology and psychiatry.
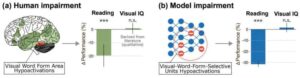
AI brain successfully mimics dyslexia and spots fonts that improve reading
For the first time, researchers have used an advanced AI model that understands both images and language, allowing them to model dyslexia, paving the way for potential new treatments.

Merck steps up as ‘meaningful competitor’ to Gilead with HIV pill approval
The newly approved HIV drug Idvynso will also help Merck diversify as loss of exclusivity looms over its top-selling product, the mega-blockbuster cancer drug Keytruda.
Amid US ordeal, Moderna wins EU approval for flu/COVID-19 combo shot
In the U.S., Moderna withdrew its approval application for the combination vaccine in May last year and the timeline for resubmission remains uncertain.
