MedTech News
.................... by Andrew Celentano

MannKind wins FDA approval for inhaled insulin for children with type 1, type 2 diabetes
MannKind (Nasdaq:MNKD) announced today that it received FDA approval for its Afrezza inhaled insulin powder for children.
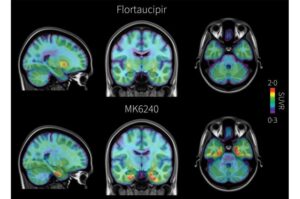
New brain scan detects Alzheimer’s tau earlier than current standard
A new brain imaging test can detect a key hallmark of Alzheimer’s disease before symptoms appear and earlier than the method currently used in clinical practice in the United States and Europe, report University of Pittsburgh School of Medicine researchers in The Lancet.
Lab-grown heart patch boosts pumping power in severe heart failure trial
Researchers at the University Medical Center Göttingen (UMG) and the University Hospital of Schleswig-Holstein (UKSH) have made a significant breakthrough in the treatment of severe heart failure: For the first time, a clinical trial has demonstrated that laboratory-grown heart muscle tissue can improve the pumping function of damaged hearts.
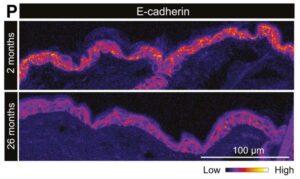
Unprecedented view inside live stem cells reveals aging process and loss of regenerative capacity
Scientists have developed a powerful new technique that allows them to observe how individual cells manufacture proteins during aging, offering an unprecedented glimpse into the hidden molecular activity of stem cells in living tissue.

Tumors May Not Develop Randomly Across the Brain — Fruit Flies May Help Explain the Pattern
Brain tumor growth may depend not only on cancer-causing mutations, but also on the molecular conditions in specific brain regions.

AbbVie snags FDA’s first ADC approval for ultra-rare blood cancer
Decnupaz is the first antibody-drug conjugate for blastic plasmacytoid dendritic cell neoplasm, an ultra-rare and aggressive blood cancer.
Procept Biorobotics wins expanded FDA IDE for Aquablation, completes study enrollment
Procept BioRobotics (Nasdaq:PRCT) announced today that it received FDA investigational device exemption (IDE) for a second Aquablation evaluation.
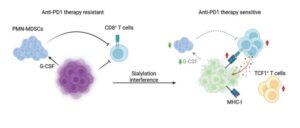
Breast tumors use sugar coating to evade immunity, opening potential immunotherapy path
An international research team led by the Medical University of Vienna has now identified a previously underestimated mechanism by which breast tumors evade the immune system.
