MedTech News
.................... by Andrew Celentano

Harnessing the Power of AI to Fight Lyme Disease
An anonymous gift helped Tufts researchers speed up a project to develop new drugs that could prevent and treat Lyme disease
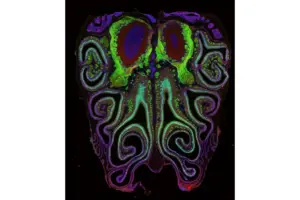
First-Ever Smell Map Is a Breakthrough in Sensory Research, a Step to Help Us Tackle Loss of Smell
Discover how smell receptors in our noses aren’t randomly arranged but are highly organized, offering new paths toward treating sensory impairment.
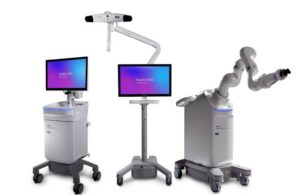
Medtronic wins CE mark for Stealth AXiS surgical robot platform
Medtronic (NYSE:MDT) announced today that it received CE mark approval for its Stealth AXiS surgical robot platform.
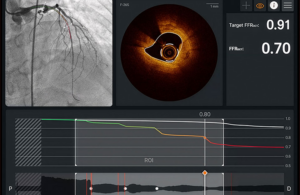
Abbott earns FDA clearance, CE mark for next-gen coronary imaging platform
Abbott (NYSE:ABT) announced today that it received FDA clearance and CE mark for its next-generation Ultreon 3.0 software.
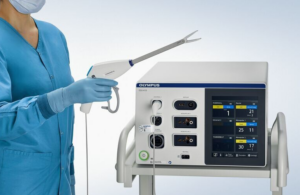
Olympus wins FDA clearance for Powerseal open extended jaw device
Olympus announced today that it received FDA 510(k) clearance for its Powerseal open extended jaw sealer/divider for use in open surgery

AI model detects normally ‘invisible’ tissue changes of pancreatic cancer at stage 0
An AI model (REDMOD) can pick up the very early subtle tissue changes of pancreatic ductal adenocarcinoma, the most common form of pancreatic cancer, which conventional imaging and the human eye find difficult to detect, finds research published online in the journal Gut.
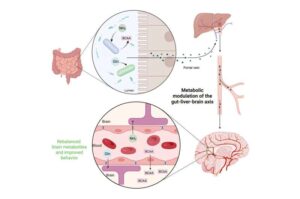
From gut to brain: Scientists engineer bacteria to treat severe liver-related brain dysfunction
A research team engineered strains of a naturally occurring beneficial gut bacterium to function as programmable therapeutics capable of restoring metabolic balance across the gut, liver and brain.
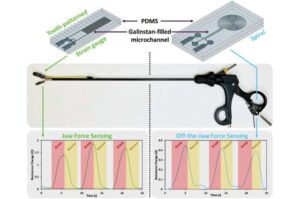
Smart soft sensors restore surgeons’ sense of touch in minimally invasive procedures
Researchers at NYU Abu Dhabi have developed soft, flexible sensors that help restore a surgeon’s sense of touch during minimally invasive (keyhole) surgery.
