MedTech News
.................... by Andrew Celentano

AI-guided robot navigates thrombectomy route from leg to brain
Researchers at King’s College London have shown for the first time that AI can autonomously perform thrombectomy navigation in a physical lab setting, a step toward expanding access to life-saving stroke treatment. For their study, now published in IEEE Robotics and Automation Letters, the team developed a robotic system that uses AI to navigate the complex pathway through blood vessels from the leg to the brain during mechanical thrombectomy (MT).
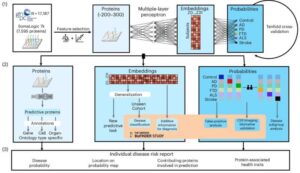
AI model can detect multiple cognitive brain diseases from a single blood sample
“Our hope is to be able to accurately diagnose several diseases at once with a single blood test in the future,” says Vogel, who led the study. He is an assistant professor, head of a research group, and part of the strategic research area MultiPark at Lund University.
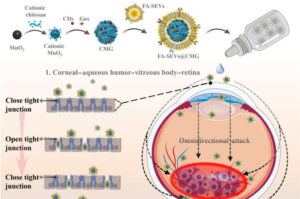
New, more effective delivery method for eye cancer treatment is derived from pig semen
Getting past the barrier surrounding the eye is a difficult but necessary part of treating retinoblastoma (RB)—a form of eye cancer that is more common in children. Once the barrier is penetrated, RB responds well to treatment. However, current methods, such as injections, chemotherapy, and radiotherapy, are painful and often cause vision loss and other serious side effects. But, a new method, described in a study published in Science Advances, might offer a safer, more effective way to treat RB.
Restore Robotics Receives FDA 510(k) Clearance for Two Additional da Vinci Xi® Robotic Instruments
HENDERSON, Nev., March 31, 2026 /PRNewswire/ — Restore Robotics today announced it has received additional 510(k) clearances from the U.S. Food and Drug Administration (FDA) for the remanufacturing of two more da Vinci Xi® robotic surgical instruments. This milestone expands Restore Robotics’ growing portfolio of cleared instruments and further strengthens its position as a leader in robotic instrument remanufacturing.

i-Lumen Scientific Receives FDA Approval of IDE to Initiate U.S. Enrollment in i-SIGHT2 Clinical Study for dry AMD
IDE approval by the FDA expands global trial to the United States as first study participant is treated in the United Kingdom

Natural Cycles integration unlocks FDA-cleared birth control app for users with select Garmin smartwatches
Garmin users can now integrate with the temperature-powered Natural Cycles app, which turns Garmin skin temperature data into fertility insights for women’s health

New FDA-Cleared At-Home Treatment Helps Men Regain Bladder Control After Prostate Surgery
NEWTOWN, Conn., March 31, 2026 /PRNewswire/ — Elidah today announced FDA clearance of Elitone for Men, a non-invasive, at-home treatment designed to help men regain bladder control after prostate surgery, addressing a significant and often overlooked men’s health condition. Urinary incontinence, or bladder leakage, is one of the most common and disruptive complications following prostate procedures, yet men have had few options to actively support recovery at home.

Breaking the PCR-Only Barrier: Dual 510(k) Clearance and CLIA-Waiver Establish FINDER as the First Molecular Point-of-Care Platform with Multifunctional Capabilities
DURHAM, N.C., March 31, 2026 /PRNewswire/ — Baebies announced FDA 510(k) clearance and CLIA-waiver approval for the FINDER® Flu A&B/SARS-CoV-2 Test, an ultra-rapid RT-PCR assay on the FINDER platform. This establishes FINDER as the first molecular point-of-care platform with multifunctional capabilities. The compact, automated system delivers PCR-quality results during the patient visit, with positives as early as 15 minutes and negatives in about 20 minutes, giving urgent care centers, emergency departments, clinics, and other decentralized settings the speed, ease of use, and reliability needed for better diagnostic decisions.
