MedTech News
.................... by Andrew Celentano
Novel blood test detects rejection after lung transplant
A simple blood test could one day replace invasive biopsies for detecting acute cellular rejection (ACR) after lung transplantation, an American Journal of Transplantation study finds.

Living bandage accelerates healing across multiple wound types
Researchers at Rice University with the support of the Rice Biotech Launch Pad have developed a cytokine factory patch designed to address this challenge by continuously producing and delivering therapeutic cytokines directly within the wound environment.
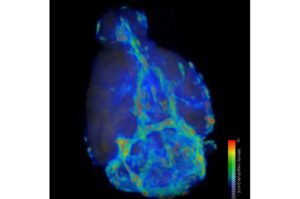
AI maps brain waste-clearing flow, revealing two speeds tied to deep sleep
Kelley and his colleagues from the University of Rochester, Brown University, and the University of Copenhagen turned to artificial intelligence for help.

FDA Breakthrough Device Designation Marks Major Milestone for Gene Solutions’ SPOT-MAS 10 Multi-Cancer Screening Test
SINGAPORE, May 27, 2026 /PRNewswire/ — Gene Solutions, a global biotechnology company advancing accessible genomic solutions for cancer detection and precision oncology, today announced that the U.S. Food and Drug Administration has granted Breakthrough Device Designation to SPOT-MAS 10, its multi-omic blood test designed to detect cancer-associated signals.

Spectral AI Receives FDA De Novo Clearance for DeepView System for Burn Indication
Spectral AI announced that the U.S. Food and Drug Administration (FDA) has granted De Novo classification for its DeepView System, authorizing the company to commence commercial distribution activities in the United States.
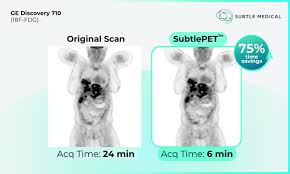
Subtle Medical Receives FDA Clearance for SubtleHD(PET) AI-Powered Image Enhancement Software
California-based Subtle Medical has announced FDA clearance for SubtleHD(PET), its next-generation PET image enhancement solution designed to improve image quality and accelerate PET imaging workflows across PET/CT and PET/MR systems.
Cardiosense Receives FDA De Novo Classification for AI-Powered Heart Failure Monitoring Software
Chicago, Illinois-based Cardiosense announced that the US Food and Drug Administration (FDA) has granted De Novo classification for its PCWP Analysis Software, which is designed to estimate noninvasively pulmonary capillary wedge pressure (PCWP), a key indicator of heart health.

A New Genetic Map of the Human Eye Could One Day Reveal Vision Loss Years Before Symptoms Start
A new map of the human eye made using donated eyeballs that could help explain and possibly prevent a handful of sight-related diseases.
