MedTech News
.................... by Andrew Celentano
Human Cells Can Pass DNA to Each Other Through Tiny Tubes — Here’s How It May Fuel Cancer Growth
DNA can be exchanged between two cells that connect by forming nanotubes, and find out why this is crucial for cancer research.

ABILITY Neurotech receives MREC approval for ALS BCI clinical trial
The clearance enables ABILITY Neurotech to move beyond intra-operative testing to long-term clinical investigation involving human participants.

Abbott wins CE mark for dual glucose-ketone sensor
Abbott (NYSE:ABT) announced today that it received CE mark approval for its dual glucose-ketone sensing technology for people with diabetes.

Wearable ultrasound patch for high-risk pregnancies could improve care
Engineers at the University of California San Diego have created a soft, wearable ultrasound patch that can continuously monitor a fetus for hours at a time—and it can do so consistently even as the fetus and umbilical cord constantly move during pregnancy.
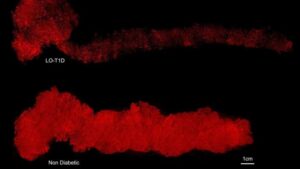
Whole organ 3D imaging reveals remaining insulin producing cells in type 1 diabetes
Researchers at Umeå University have conducted a unique three-dimensional mapping of an entire human pancreas. The study shows that insulin-producing cells can remain long after the onset of type 1 diabetes—a finding that suggests the disease progression is more complex than previously assumed.
FDA Grants Breakthrough Device Designation to TOBY Urine-Based Test for Alzheimer’s Disease
The U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation to the TOBY Alzheimer’s Test, a urine-based diagnostic test developed by TOBY, a biotechnology company based in Austin, Texas.
CSR secures certifications in EU and Singapore for Sentire system
The CE certification confirms compliance with regulatory standards for safety, efficacy, and quality management.
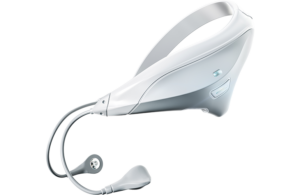
Neurovalens earns FDA nod for neuromod for PTSD
Neurovalens announced today that it received FDA de novo approval for Modius Spero, a post-traumatic stress disorder (PTSD) therapy.
