
CroíValve reports positive first-in-human valve repair data
CroíValve today announced favorable patient outcomes in a first-in-human clinical trial of its Duo tricuspid coaptation valve system.

CroíValve today announced favorable patient outcomes in a first-in-human clinical trial of its Duo tricuspid coaptation valve system.

The approval allows women to self-collect vaginal specimens for HPV testing in a health care setting, which could include non-traditional locations such as a retail pharmacy or mobile clinic.

Carthera announced today that it received FDA and European orphan drug designation (ODD) for the use of carboplatin with its SonoCloud.

Medtronic (NYSE: MDT)+
announced today that its PulseSelect pulsed field ablation (PFA) system has won regulatory approval in Japan.
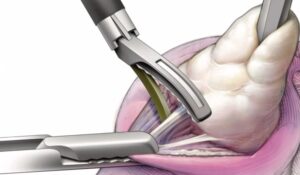
Freyja Healthcare Brings First-of-Its-Kind 2mm Abdominal-Access Device to Laparoscopic Surgery to Further Innovation in Fast-Growing Women’s Health Market
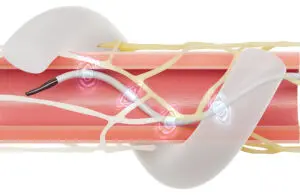
Medtronic (NYSE: MDT)+
today announced its Symplicity Spyral renal denervation system won National Medical Products Administration (NMPA) approval in China.
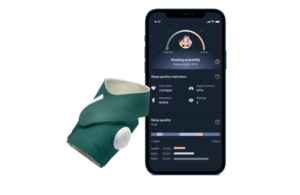
Owlet announced that it received CE mark approval for its Dream Sock device for monitoring the vital signs of infants.

Lightning Flash 2.0 features advanced computer assisted vacuum thrombectomy (CAVT) software, designed for increased efficiency and sensitivity to enhance removal of venous thrombus and treatment of pulmonary emboli (PE)
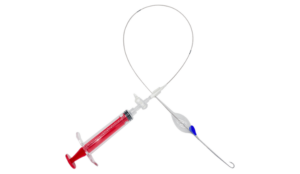
Approval grants EU providers access to crucial life-saving device, indicative of the company’s global growth and ability to increase emergency and trauma care.

Monteris Medical today said its launch of the NeuroBlate NB3 FullFire makes it the smallest laser probe for brain procedures on the market.