
FDA clears new mammography system from Siemens Healthineers
Siemens Healthineers announced today that it received FDA 510(k) clearance for its MammoMat B.brilliant mammography platform.

Siemens Healthineers announced today that it received FDA 510(k) clearance for its MammoMat B.brilliant mammography platform.
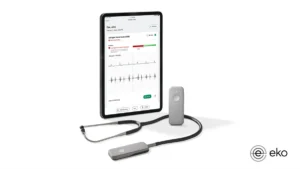
The clearance adds to the list of devices the FDA has authorized this year with AI algorithms to detect health conditions.

The CEO of Prenosis told MedTech Dive the company sees third-party validation as important, with the FDA having clarified that certain decision support tools should be regulated as medical devices.

TEG testing provides critical information that can help physicians improve hemostasis management for their patients

HeadaTerm 2 uses neuromodulation technology. It releases targeted electrical impulses that increase the pain tolerance of the wearer.
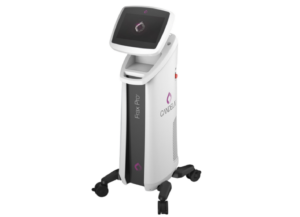
The Venus Versa Pro combines the applicator of the Venus Viva MD with the Venus Versa system which are both approved in Australia and registered in Australian Register of Therapeutic Goods (ARTG).

This milestone gives healthcare professionals an important tool for managing bacteremia by providing antibiotic susceptibility test (AST) results with unprecedented speed.

Benefits of the system, according to a news release, include maneuverability and visualization.
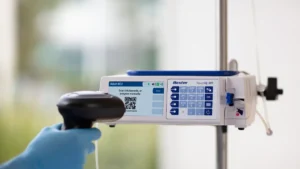
The clearance ends a three-year back-and-forth with the FDA to get the product to market.
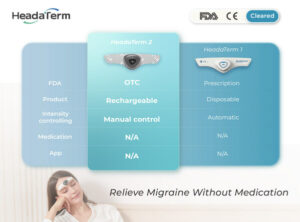
Using neuromodulation technology, HeadaTerm 2 releases targeted electrical impulses to increase pain tolerance of its users. It is FDA-cleared for the preventative treatment of migraine headaches.