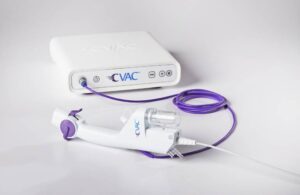
Calyxo Announces FDA Clearance for New, Redesigned CVAC System
New Solution for Kidney Stone Treatment Already Used to Treat More than 50 Patients

New Solution for Kidney Stone Treatment Already Used to Treat More than 50 Patients

Medical imaging AI company receives approval for algorithms enabling incidental detection of pulmonary embolism and automatic assessment of stroke severity.
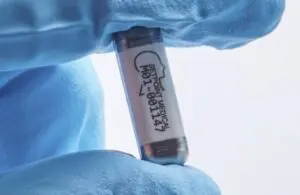
SetPoint Medical announced today that the FDA accepted it into the Total Product Life Cycle Advisory Program (TAP) pilot.

Sequel Med Tech will sell the new system, which integrates with Tidepool’s Loop insulin dosing algorithm.

Solution uses fully automated AI segmentation to generate inHEART’s digital twin of the heart with greater speed and efficiency

The soft tissue robotics leader incorporated new features into the system, from tissue sensing feedback to a smaller size and more comfortable console.

Inspectors found fault with the handling of corrective and preventive actions for the Ivenix Infusion System.

Axonics (Nasdaq:AXNX) announced today that it received CE mark approval for its R20 rechargeable sacral neuromodulation (SNM) system.

SafeGuard Surgical announced today that the FDA granted breakthrough device designation to its LeakGuard biodegradable stent.
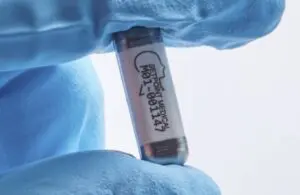
SetPoint Medical announced today that received FDA breakthrough device designation for its novel neuroimmune modulation platform.