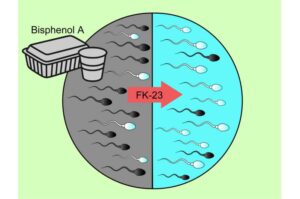
Heat-treated probiotic may protect sperm from BPA-linked damage, rat study suggests
A research team led by Visiting Professor Yukiko Minamiyama of the Graduate School of Medicine at OMU investigated whether a paraprobiotic material derived from the lactic acid bacterium Enterococcus faecalis (known as FK‑23) protects against the sperm toxicity caused by BPA.