MedTech News
.................... by Andrew Celentano
For the First Time, Lab‑Grown Brain Organoids Display Ability to Learn
Learn more about the advances in brain organoids and what this science could mean for the future.
New insights into a hidden process that protects cells from harmful mutations
Research reveals how cells may activate a compensation system that can reduce the effects of harmful genetic mutations. This could inform gene therapy development.
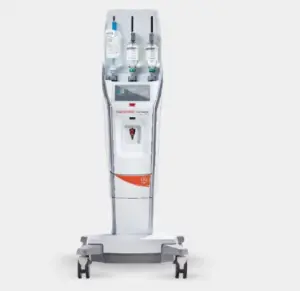
Bayer secures expanded FDA clearances for MRI contrast agent injector
The FDA’s expanded clearances broaden the application of Bayer’s Medrad MRXperion system across a wider range of MR settings.
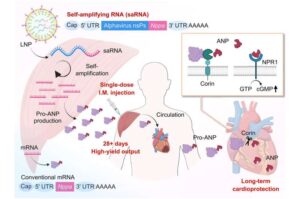
Single saRNA shot helps with healing after a heart attack
This simple injection may one day help people recover more safely and fully after a heart attack.
Scientists engineer ‘living eye drop’ to support corneal healing
University of Pittsburgh School of Medicine researchers have developed an early-stage, experimental “living eye drop” that uses naturally occurring eye bacteria to support corneal wound healing.
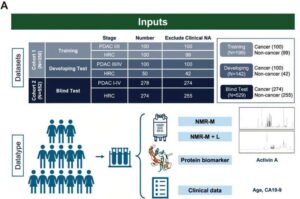
AI blood test detects early pancreatic cancer with up to 94% accuracy
A team of researchers from Taiwan has developed PanMETAI, an AI-powered platform that analyzes metabolic fingerprints in a simple blood sample to detect pancreatic cancer at its earliest stages—when treatment is most effective—achieving up to 94% diagnostic accuracy.
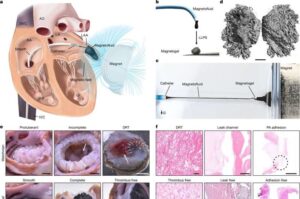
A safer, more effective atrial fibrillation treatment method using magnetic gel may be on the way
Researchers have developed a promising new treatment involving magnetic fluids.
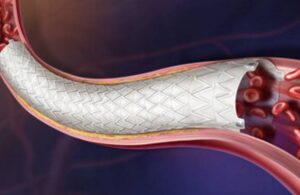
BD picks up CE mark for Revello vascular covered stent
BD (NYSE: BDX)+ announced today that it received CE mark approval for its Revello vascular covered stent system.
