MedTech News
.................... by Andrew Celentano

How neurons sense bacteria in the gut
Neural interaction with bacteria has important effects on animal brains. A new study investigates how neurons sense bacteria by revealing, in nematodes, the bacterial signals that a key neuron detects.
Treatment of rare childhood epilepsy could begin before birth
RNA therapy may calm abnormal brain activity of epilepsy in utero, before seizures emerge
enVVeno Medical secures FDA IDE approval for venous valve study
The TAVVE study will begin later this year with ten patients, whose 30-day safety results will be submitted to the FDA.

New tool launches to support women through post-loss journey
https://med-techinsights.com/2026/04/22/new-tool-launches-to-support-women-through-post-loss-journey/
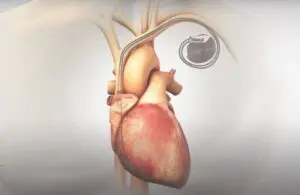
Orchestra BioMed wins FDA breakthrough nod for pacemaker in hypertension
Orchestra BioMed (Nasdaq:OBIO) announced today that it received a second FDA breakthrough device designation for its AVIM therapy.
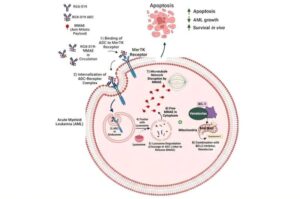
Targeted ‘biological missile’ blocks leukemia growth while sparing healthy tissue
New research out of VCU Massey Comprehensive Cancer Center has uncovered a targeted cancer therapy that significantly prevents leukemia progression, improves survival rates and minimizes damage to surrounding healthy tissue.
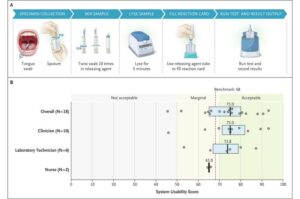
A single swab paves the way for simplified tuberculosis diagnosis
Led by researchers from Heidelberg Faculty of Medicine at Heidelberg University and the University of California, San Francisco (U.S.), an international team has evaluated a novel approach for the diagnosis of tuberculosis.
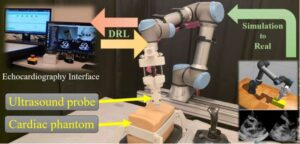
New AI-powered robotic system performs heart ultrasounds without guidance
A Concordia-led team of researchers has developed a new AI-driven robotic system that can perform cardiac ultrasound scans autonomously. The researchers say this approach could expand access to cardiac imaging in remote or underserved areas, reduce operator fatigue, and standardize scan quality. The work is published in the journal IEEE Transactions on Medical Robotics and Bionics.
